7. Energy, Rate and Equilibrium
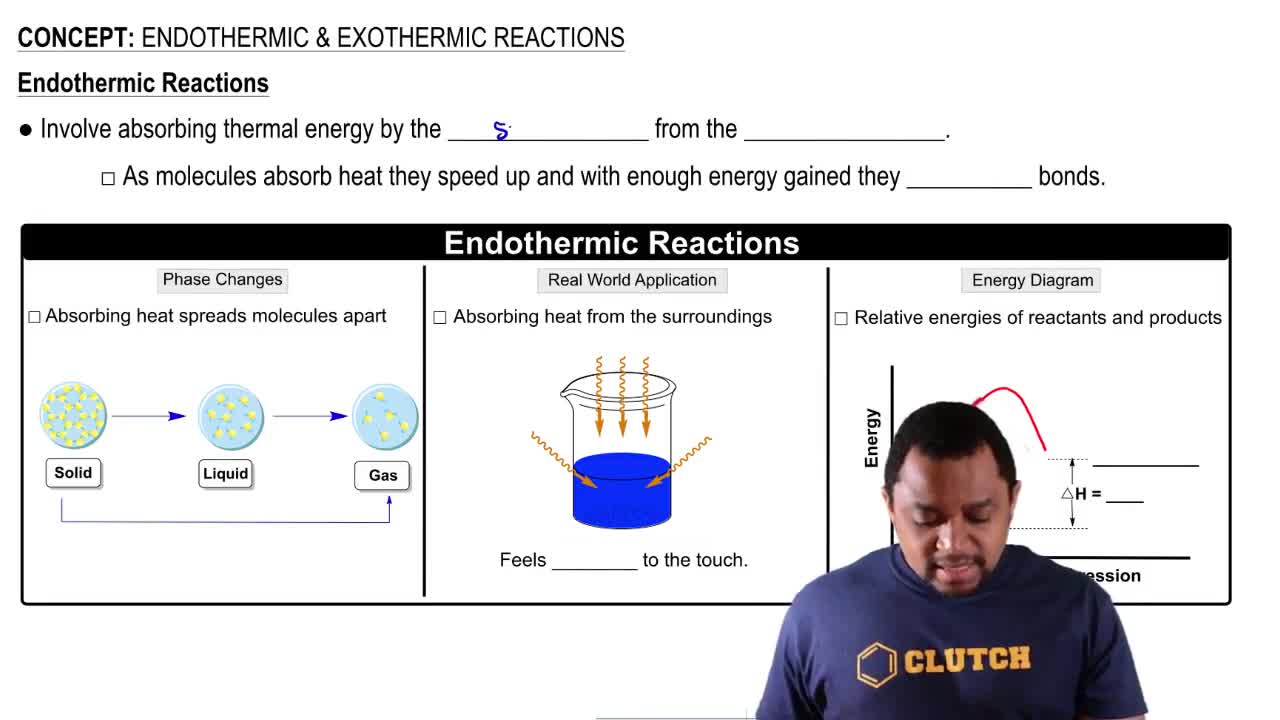
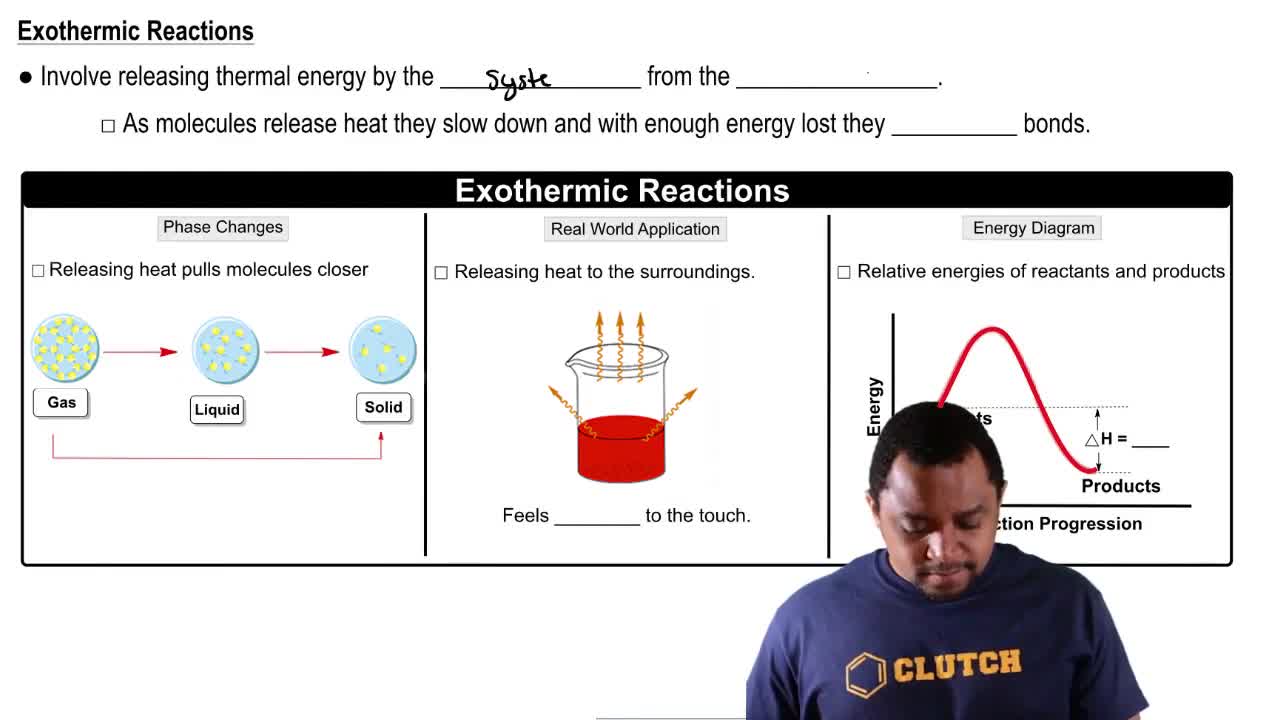
Endothermic & Exothermic Reactions
Problem 107
Textbook Question
Textbook QuestionThe equation for the formation of silicon tetrachloride from silicon and chlorine is (7.9) Si(s) + 2Cl₂(g) → SiCl₄(g) + 157 kcal b. Is the energy of the product higher or lower than the energy of the reactants?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
863
views
Was this helpful?
Related Videos
Related Practice