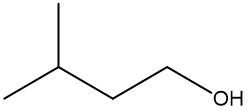
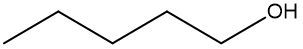
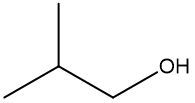
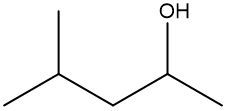
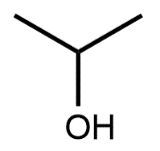
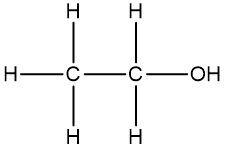
In this video, we're going to take a look at skeletal formulas. Now, this is a representation, also known as a bond line or Line Formula, and it's the fastest way of drawing complex organic structures. As we go deeper and deeper into organic chemistry, we're going to move away from our structural formulas and condensed formulas because they take too long to draw. We're going to start incorporating more line, bond line, or skeletal formulas to draw more complex organic compounds. Now, here we have our structural formula which shows the connectivity between different atoms. We can package everything in more tightly, so it becomes CH3 CH2OH. This can be transformed into our skeletal formula. Now before we dissect it, let's go over a few guidelines here.
First, Carbon-Carbon Bonds are shown as lines, where every corner represents a carbon atom with enough hydrogen atoms. So, this here is a carbon, and this here is a carbon. And then if we continue, we land at the oxygen because there's an oxygen right here. Now, carbon and hydrogen atoms are not shown but other atoms such as oxygen, nitrogen, and sulfur are shown. So here, if we look, we have an oxygen right there. Then, we're going to say there's another exception to this, and that exception is hydrogen atoms attached to atoms other than carbon are shown. So if we look, we show this oxygen, and we show the hydrogen connected to it.
Now, if we take a look here, we're going to say we know that these two are carbons; carbon must make 4 bonds. So, this carbon here on the left, we see it making one bond to this carbon in the middle. It needs to make 4 bonds, so its 3 hydrogens aren't there, they're just invisible. The carbon in the middle, that carbon we see it making 2 bonds. Remember, carbon must make 4 bonds, so it has 2 hydrogens that are invisible. So, as we can see, without having to show all of this, I can just do this Skeletal Formula, and it represents the Condensed Formula, and it represents the Structural Formula. Again, Skeletal Formula is just a quick, faster way to draw these more complex organic compounds, and we're going to utilize them more and more as we delve deeper into organic chemistry.