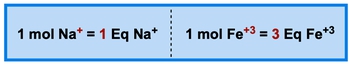
Equivalents are essential for quantifying the amount of individual ions in body fluids and intravenous solutions. An equivalent, often abbreviated as eq, represents the number of moles of charge contributed by an ion in a solution. Specifically, one equivalent corresponds to one mole of positive or negative charge. It is crucial to note that equivalents can only be expressed as positive values.
For instance, when considering sodium ions (Na+), one mole of sodium contributes a charge of +1, resulting in 1 equivalent. In contrast, for iron (III) ions (Fe3+), one mole contributes a charge of +3, which translates to 3 equivalents. To calculate the number of equivalents for any ion, the formula is straightforward: multiply the ion's charge by the number of moles of that ion present.
Additionally, the term milliequivalent is commonly used to express equivalence in smaller units, where 1 equivalent equals 1,000 milliequivalents. Thus, the relationship can be summarized as follows: equivalent = ion charge × moles of ions. For example, with sodium ions, the charge is +1 and the number of moles is 1, yielding 1 equivalent. For iron (III) ions, the charge is +3 with 1 mole present, resulting in 3 equivalents. This understanding is vital when calculating the equivalence of any ionic solution.