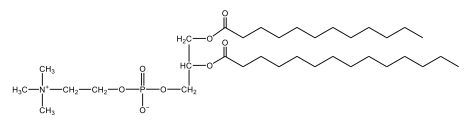
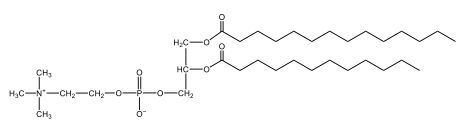
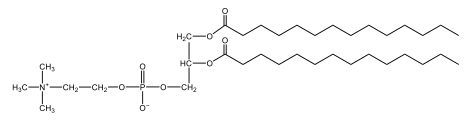
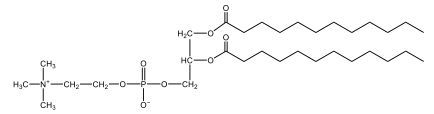
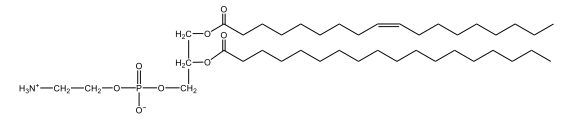
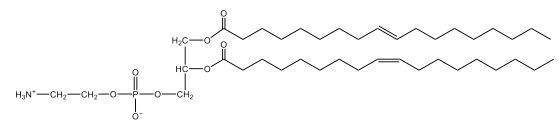
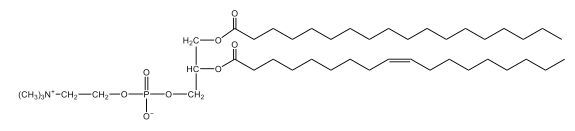
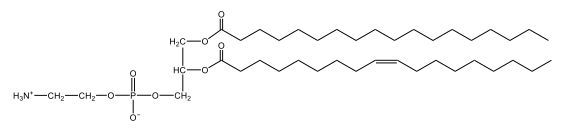
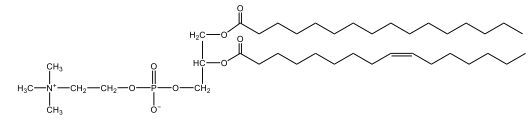
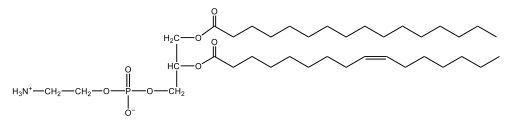
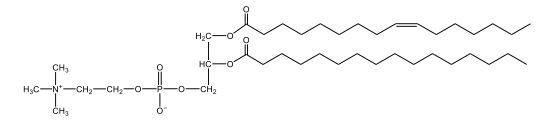
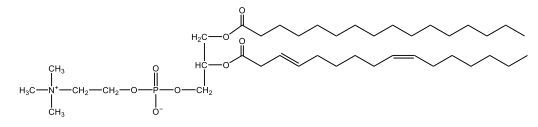
Phospholipids are a specific type of lipid characterized by the presence of a phosphate group attached to either a glycerol or sphingosine backbone. These molecules are amphipathic, meaning they possess both a hydrophilic (water-attracting) head and a hydrophobic (water-repelling) tail. The hydrophilic head is polar, while the hydrophobic tail is non-polar. This unique structure allows phospholipids to play a crucial role in biological systems.
In the broader classification of lipids, they can be divided into fatty acids and steroids. Focusing on fatty acids, they can further be categorized into waxes, glycerolipids, sphingolipids, and eicosanoids. Phospholipids fall under the categories of glycerolipids and sphingolipids due to their phosphate group attachment.
When phospholipids are placed in an aqueous environment, they naturally orient themselves to form a lipid bilayer. In this arrangement, the polar heads face outward towards the water, while the hydrophobic tails are tucked away in the interior. This structural organization is vital as it forms the fundamental basis of all cell membranes, highlighting the importance of phospholipids in cellular architecture and function.