13. Liquids, Solids & Intermolecular Forces
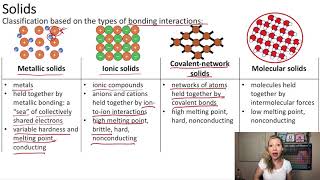
Atomic, Ionic, and Molecular Solids
Practice this topic
- Multiple Choice
What is the major electrostatic force found between ammonia molecules, NH3?
1788views3rank2comments - Multiple Choice
As it cools off, olive oil slowly hardens and forms a solid over a range of temperatures. Which best describes it as a solid?
1584views8rank2comments - Multiple Choice
Compound A is hard, doesn't conduct electricity, and melts at 1400ºC. Compound A represents which of the following:
1361views8rank1comments - Multiple Choice
Classify each solid as amorphous, molecular, network covalent, alloy or ionic.
a) Steel ______________________
b) CO2 ______________________
c) Graphite ______________________
d) CaCO3 ______________________
e) Bronze, an alloy of Cu and Sn ______________________
1918views5rank - Textbook Question
Shown here are sketches of two processes. Which of the processes refers to the ductility of metals and which refers to malleability of metals? (a)
(b)
300views - Textbook QuestionThe following diagrams represent the electron population ofthe composite s–d band for three metals—Ag, Mo, and Y:Which diagram corresponds to which metal? (LO 12.7)(a) Ag = 3, Mo = 1, Y = 2(b) Ag = 2, Mo = 1, Y = 3(c) Ag = 2, Mo = 3, Y = 1(d) Ag = 1, Mo = 2, Y = 3344views
- Textbook QuestionExamine diagrams for the electron population of the composite s–d band for three metals in question 6. Which metal has the highest melting point? (LO 12.7) (a) Metal 1 (b) Metal 2 (c) Metal 3361views
- Textbook Question
At three different temperatures, T1, T2, and T3, the molecules in a liquid crystal align in these ways:
(a) At which temperature or temperatures is the substance in a liquid crystalline state? At those temperatures, which type of liquid crystalline phase is depicted?
594views1rank