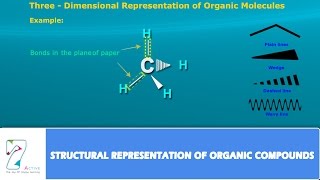
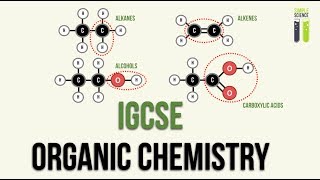
22. Organic Chemistry
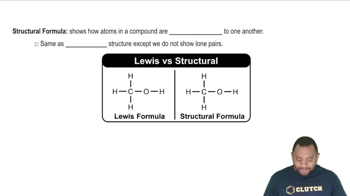
Structural Formula
22. Organic Chemistry
Structural Formula
Additional 4 creators.
Learn with other creators
Showing 7 of 7 videos
Practice this topic
- Multiple ChoiceButane and methylpropane are examples of _________.1189views
- Multiple ChoiceThe type of isomerism in alkenes that is due to restricted rotation is1043views
- Multiple Choice
Propane (C3H8) is a component of liquified petroleum gas. Draw a structural formula for propane. (Hint: Carbon makes four bonds.)
1181views3rank - Multiple Choice
Methylamine (CH5N) is a gaseous compound with a fishy smell. Draw this molecule using structural formula.
911views1rank1comments - Open Question
The type of formula that shows the arrangements of atoms and bonds is called -
694views