10. Periodic Properties of the Elements
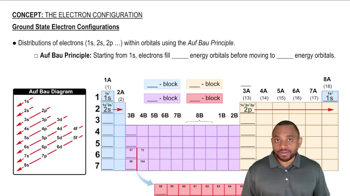
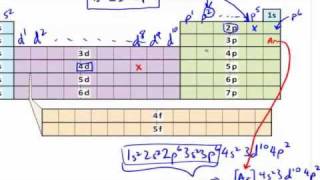
The Electron Configuration
10. Periodic Properties of the Elements
The Electron Configuration
Showing 6 of 6 videos
Additional 6 creators.
Learn with other creators
Showing 9 of 9 videos
Practice this topic
- Multiple Choice
Which electron configuration represents a violation of Hund's Rule?
5319views34rank1comments - Multiple Choice
Which electron configuration represents a violation of the Auf Bau Principle?
3838views15rank - Multiple Choice
Identify the element with the given electron orbital diagram.
5477views15rank - Multiple Choice
Write the electron configuration and electron orbital diagram for the following element:
Mn (Z = 25)
2782views14rank2comments - Open Question
Use the periodic table to write the electron configuration of selenium (Se).
1133views - Open Question
Give the complete ground-state electron configuration for silicon (Si).
1141views - Open QuestionWrite the electron configuration of the following elements913views
- Open QuestionWhich electron configuration represents a violation of the pauli exclusion principle?1467views1rank