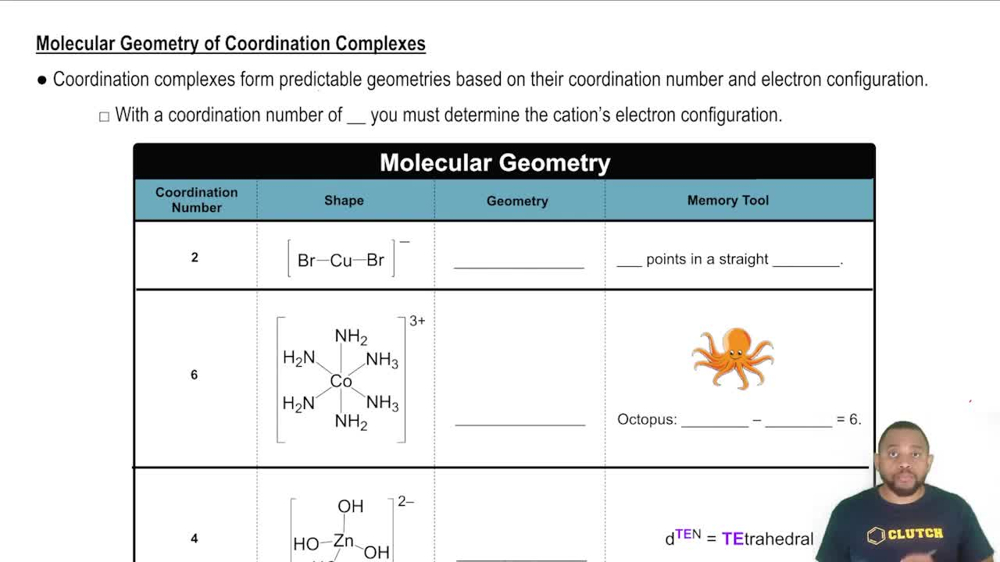
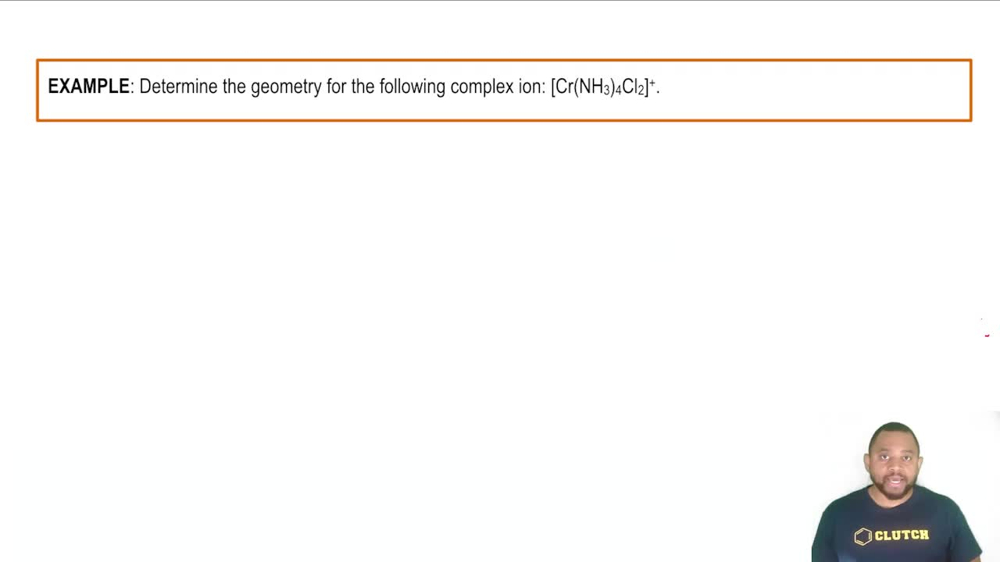
24. Transition Metals and Coordination Compounds
Coordination Numbers & Geometry
Practice this topic
- Multiple Choice
Correctly label all the components of the coordination complex and its coordination number: Na2[SnCl6].
611views - Multiple Choice
Determine the geometry for the following complex ion: [Ni(OH)4]2–.
734views - Multiple Choice
Determine the geometry for the following complex molecule: Pd(H2O)4.
687views - Textbook QuestionIn excess of NH3(aq), Zn2+ forms a complex ion, [Zn(NH3)4]2+ which has a formation constant Kf = 7.8 x 10^8. Calculate the concentration of Zn2+ in a solution prepared by adding 1.00 x 10^-2 mol Zn(NO3)2 to 1.00 L of 0.250 M NH3. (a) 7.9 x 10^-4 M(b) 2.8 x 10^-6 M(c) 3.9 x 10^-9 M(d) 6.4 x 10^-11 M2280views
- Textbook Question
Draw the structure of the iron oxalate complex [Fe(C2O4)3]3-. Describe the coordination geometry, and identify any chelate rings. What are the coordination number and the oxidation number of the iron?
120views - Textbook Question
Draw the structure of the following complexes. What are the oxidation state, coordination number, and coordination geometry of the metal in each?
(a) Na[Au(CN)2]
95views - Textbook Question
Draw the structure of the following complexes. What are the oxidation state, coordination number, and coordination geometry of the metal in each?
(b) [Cr(NH3)2(C2O4)2]NO2
112views - Multiple ChoiceWhat is the coordination number of the complex ion [Co(en)_2(C_2O_4)]^+?391views