22. Organic Chemistry
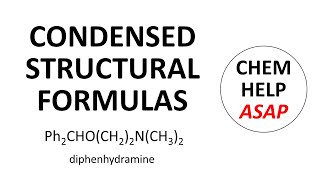
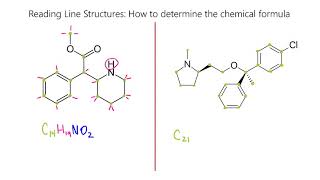
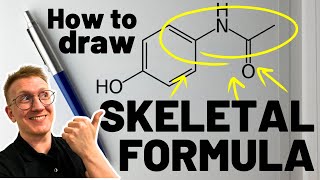
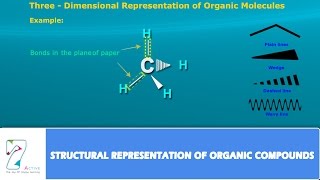
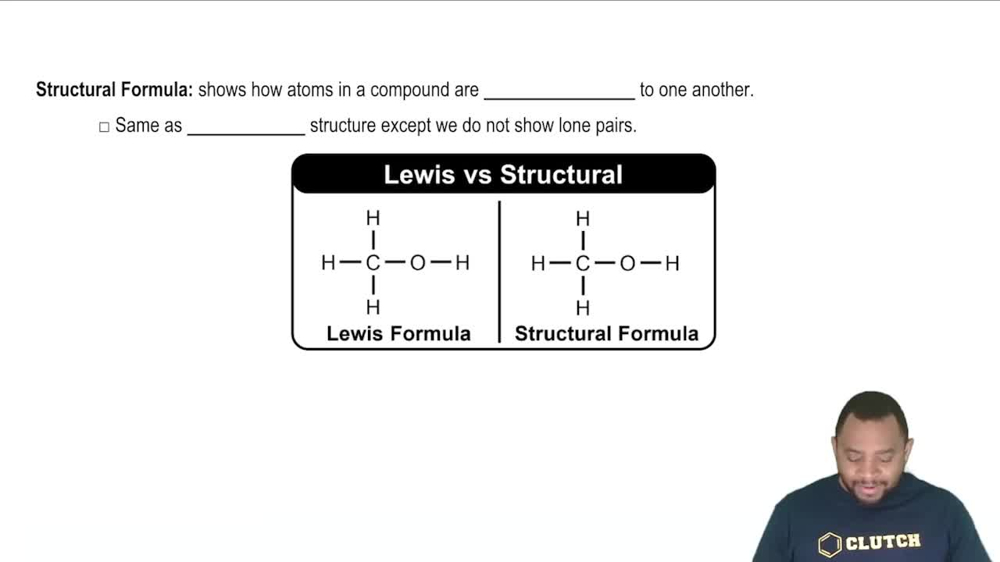
Structural Formula
Learn with other creators
Practice this topic
- Multiple ChoiceButane and methylpropane are examples of _________.1429views
- Multiple ChoiceThe type of isomerism in alkenes that is due to restricted rotation is1263views
- Multiple Choice
Propane (C3H8) is a component of liquified petroleum gas. Draw a structural formula for propane. (Hint: Carbon makes four bonds.)
1411views3rank - Multiple Choice
Methylamine (CH5N) is a gaseous compound with a fishy smell. Draw this molecule using structural formula.
1140views1rank1comments - Textbook Question
Butane, the fuel used in disposable lighters, has the formula C4H10. The carbon atoms are connected in the sequence C-C-C-C, and each carbon has four covalent bonds. Draw the structural formula of butane.
97views - Textbook QuestionCyclohexane, C6H12, is an important starting material used in the industrial synthesis of nylon. Each carbon has four covalent bonds, two to hydrogen and two to other carbons. Draw the structural formula of cyclohexane.1019views
- Textbook Question
Isooctane, the substance in gasoline from which the term octane rating derives, has the formula C8H18. Each carbon has four covalent bonds, and the atoms are connected in the sequence shown. Draw the complete structural formula of isooctane.
631views - Open Question
The type of formula that shows the arrangements of atoms and bonds is called -
898views