11. Bonding & Molecular Structure
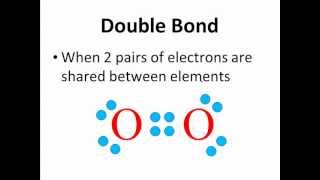
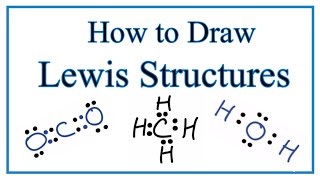
Lewis Dot Structures: Neutral Compounds
11. Bonding & Molecular Structure
Lewis Dot Structures: Neutral Compounds
Additional 4 creators.
Learn with other creators
Showing 7 of 7 videos
Practice this topic
- Multiple Choice
Determine the Lewis Dot Structure for the disulfur dichloride molecule, S2Cl2.
4506views11rank3comments - Multiple Choice
How many lone pairs are on the central element for the following compound:AsH3.
2357views10rank2comments - Multiple Choice
How many total bonding electrons are on the central element for the following compound:CO2.
1755views12rank5comments - Multiple Choice
How many lone pairs are on the central element for the following compound:NOCl
2104views5rank3comments - Open Question
Determine the Lewis Dot Structure for the diazene molecule, N2H2.
3163views29rank2comments - Open Question
Complete these structures by adding electrons, in the form of dots, as needed.
873views - Open QuestionChoose a likely identity for x, y, and z in these structures.1119views
- Open QuestionWhich molecule has only one lone (non-bonding) pair in its lewis model?1189views