11. Bonding & Molecular Structure
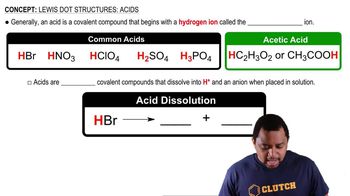
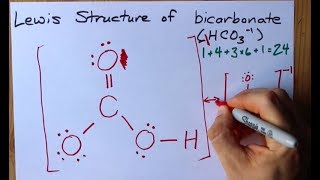
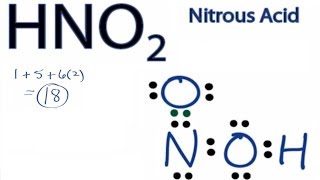
Lewis Dot Structures: Acids
11. Bonding & Molecular Structure
Lewis Dot Structures: Acids
Additional 3 creators.
Learn with other creators
Showing 6 of 6 videos
Practice this topic
- Multiple Choice
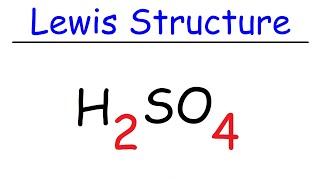
Determine the Lewis Dot Structure for the following acid:H2SO4
3609views4rank - Multiple Choice
Determine the Lewis Dot Structure for hydrocyanic acid:HCN
1815views5rank1comments - Multiple Choice
Determine the Lewis Dot Structure for chloric acid:HClO3
5207views2rank3comments - Multiple Choice
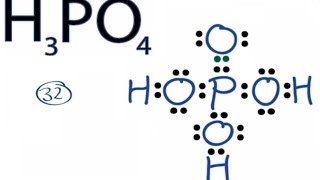
Determine the Lewis Dot Structure for phosophoric acid:H3PO4
4112views5rank1comments - Open Question
Draw a lewis structure for so2 in which all atoms obey the octet rule. Show formal charges. Do not consider ringed structures.
881views - Open Question
Draw a Lewis structure for SO2 in which all atoms obey the octet rule. Show formal charges. Do not consider ringed structures.
845views - Open Question
In molecules, C, H, O, and N atoms usually make __, __, __, and __ bonds respectively.
886views - Open Question
Determine the number of valence electrons in HCN and then draw the corresponding Lewis structure.
862views