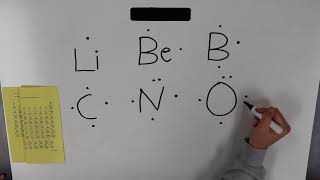11. Bonding & Molecular Structure
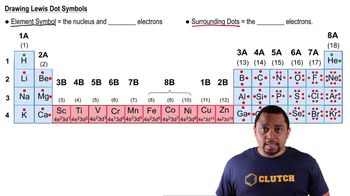
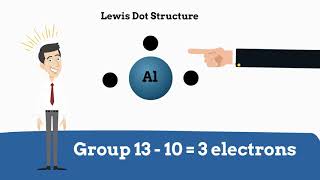
Lewis Dot Symbols
11. Bonding & Molecular Structure
Lewis Dot Symbols
Additional 3 creators.
Learn with other creators
Showing 6 of 6 videos
Practice this topic
- Multiple Choice
Draw the Lewis Dot symbol for the following ion:Co+
1906views22rank - Multiple Choice
Draw the Lewis Dot symbol for the following ion: Cd2+
2321views23rank1comments - Multiple Choice
Draw the electron-dot symbol for the following ion:P3–
2698views18rank1comments - Multiple ChoiceHow many unpaired electrons are in the Lewis symbol of an oxygen atom?850views
- Open Question
In ionic bonds will the alkali metals lose or gain electrons, how many?
766views - Open Question
Which particles are represented by the dots in Lewis valence e-dot structures?
936views - Open Question
How many covalent bonds does carbon form if each of its unpaired electrons participates in one bond?
854views - Open Question
How many covalent bonds does carbon form if each of its unpaired electrons participates in one bond?
621views