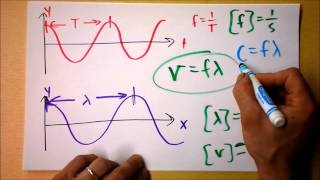
9. Quantum Mechanics
Speed of Light
Learn with other creators
Practice this topic
- Multiple Choice
Even the music we listen to deals with how energy travels to get to our car radio. If an FM Radio station broadcasts its music at 97.7 MHz find the wavelength in angstroms of the radio waves. One angstrom is equal to 10-10 m.
2050views15rank - Multiple Choice
The distance between the earth and the sun is 1.496 x 1017 μm. How long (in mins) will it take for light to go from the sun to earth?
2490views10rank - Multiple Choice
When they are burned, certain elements emit light at a specific wavelength. Some wavelengths for certain elements are provided below:
When burned, an unknown element emits light at a frequency of 9.23 x 1014 s-1. What is the identity of this unknown element?
2584views11rank - Open Question
An FM radio station found at 103.1 on the FM dial broadcasts at a frequency of 1.031 × 108 s−1 (103.1 MHz). What is the wavelength of these radio waves in meters?
881views - Open Question
Calculate the wavelength (in nm) of the blue light emitted by a mercury lamp with a frequency of 6.88 × 1014 Hz.
1466views - Open Question
The wavelength of some red light is 700.5 nm. what is the frequency of this red light?
880views - Open Question
Calculate the wavelength (in nm) of the red light emitted by a neon sign with a frequency of 4.74 × 1014 Hz.
876views - Multiple ChoiceHow much time (in seconds) does it take light to travel 1.20 billion km?511views