9. Quantum Mechanics
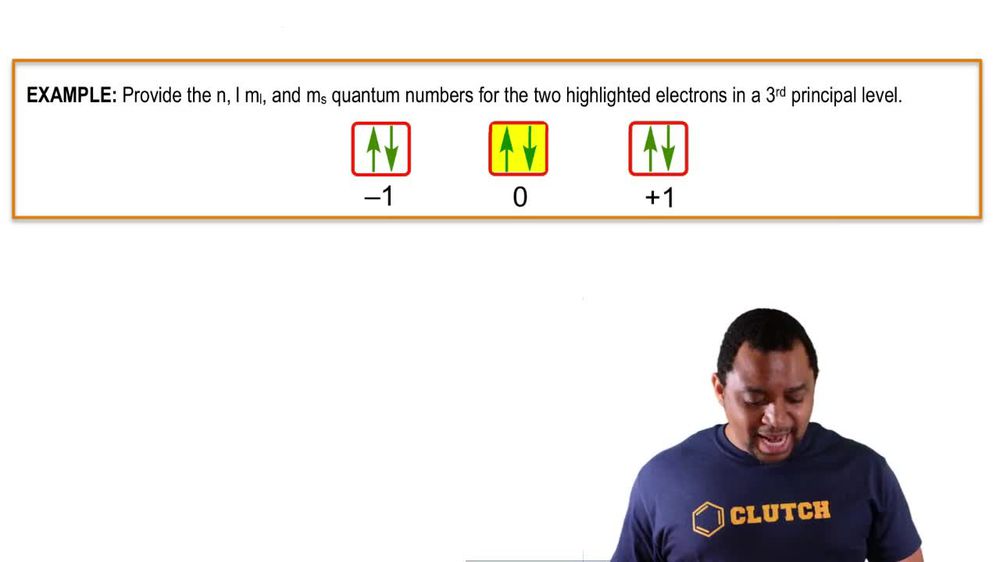
Quantum Numbers: Spin Quantum Number
Practice this topic
- Multiple Choice
Select the correct quantum numbers for the highlighted electrons in a set of 5d orbitals.
1604views4rank - Multiple Choice
Which of the following set of quantum numbers is possible?
2162views6rank - Multiple Choice
Which of the following set of quantum numbers is possible for an electron in a set of 6f orbitals?
1880views6rank - Multiple Choice
Which of the following statements is false?
1905views4rank - Textbook Question
A certain orbital of the hydrogen atom has n = 4 and l = 3. (b) What are the possible values of ms for the orbital?
560views - Textbook QuestionGive the allowable combinations of quantum numbers for each of the following electrons.(a) A 4s electron(b) A 3p electron(c) A 5f electron(d) A 5d electron627views
- Textbook QuestionAssign a set of four quantum numbers to each electron in oxygen.534views
- Textbook Question
Determine whether each of the following sets of quantum numbers for the hydrogen atom are valid. If a set is not valid, indicate which of the quantum numbers has a value that is not valid: (a) n = 3, l = 3, ml = 2, ms = +1/2 (b) n = 4, l = 3, ml = -3, ms = +1/2 (c) n = 3, l = 1, ml = 2, ms = +1/2 (d) n = 5, l = 0, ml = 0, ms = 0 (e) n = 2, l = 1, ml = 1, ms = -1/2
1456views