9. Quantum Mechanics
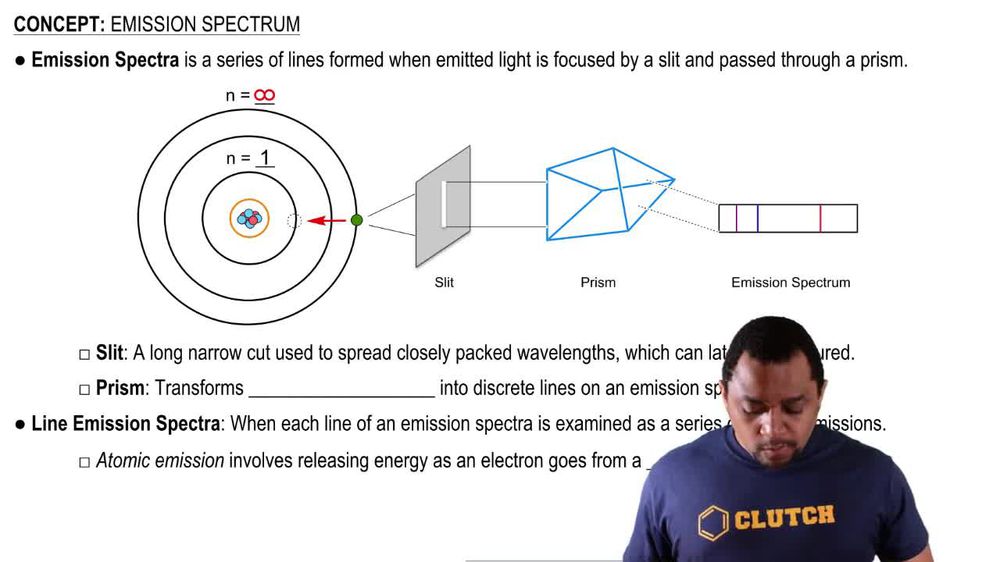
Emission Spectrum
Learn with other creators
Practice this topic
- Multiple ChoiceConsider the mass spectrum for a hypothetical element given below.
Which of the following statements correctly relates to this spectrum?897views - Multiple ChoiceWhen an electron in a hydrogen atom moves from n = 8 to n = 2, light with what wavelength is emitted?1200views
- Multiple ChoiceWhich of the following transitions results in the emission of the highest-energy photon?1014views
- Multiple ChoiceHow many photons of light are emitted in a 4.50-mJ pulse of a laser with a wavelength of 555 nm?904views
- Textbook Question
When a copper salt such as Cu(NO3)2 is burned in a flame, a blue-green color is emitted. Which figure represents the emission spectrum for the element copper? (LO 5.6)? (a)
(b)
(c)
(d)
1133views1rank - Textbook Question
A certain quantum-mechanical system has the energy levels shown in the accompanying diagram. The energy levels are indexed by a single quantum number n that is an integer. (b) Which quantum numbers are involved in the transition that requires the least energy?
589views - Textbook Question
The visible emission lines observed by Balmer all involved nf = 2. (a) Which of the following is the best explanation of why the lines with nf = 3 are not observed in the visible portion of the spectrum: (i) Transitions to nf = 3 are not allowed to happen, (ii) transitions to nf = 3 emit photons in the infrared portion of the spectrum, (iii) transitions to nf = 3 emit photons in the ultraviolet portion of the spectrum, or (iv) transitions to nf = 3 emit photons that are at exactly the same wavelengths as those to nf = 2.
1104views - Textbook Question
The Lyman series of emission lines of the hydrogen atom are those for which nf = 1. (a) Determine the region of the electromagnetic spectrum in which the lines of the Lyman series are observed.
657views