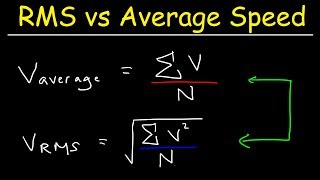7. Gases
Root Mean Square Speed
7. Gases
Root Mean Square Speed
Showing 9 of 9 videos
Practice this topic
- Multiple Choice
Determine which gas would have a root mean square speed of 515.59 m/s at 405 K.
1340views2rank - Multiple Choice
The root mean square speed of gas molecules is 283.0 m/s at a given temperature T when the recorded molar mass is 42.0 g/mol. What would be the root mean square speed for a gas with a molar mass of 152.0 g/mol?
2060views2rank2comments - Multiple ChoiceCalculate the root mean square velocity of nitrogen molecules at 20.0℃.1232views
- Textbook QuestionThe coldest temperature recorded at ground level on Earth was -89.2 °C at the Vostok Station in Antarctica. What is the speed of a nitrogen molecule at this temperature? (LO 10.9)(a) 933 m/s (b) 40.2 m/s(c) 404 m/s (d) 12.8 m/s740views
- Textbook QuestionRadon (Rn) is the heaviest (and only radioactive) member ofthe noble gases. How much slower is the root-mean-squarespeed of Rn than He at 300 K?1688views
- Textbook Question
The temperature of a 5.00-L container of N2 gas is increased from 20 °C to 250 °C. If the volume is held constant, predict qualitatively how this change affects the following: (b) the rootmean-square speed of the molecules. (c) the strength of the impact of an average molecule with the container walls. (d) the total number of collisions of molecules with walls per second.
755views - Textbook Question
(b) Calculate the rms speed of NF3 molecules at 25 °C.
1243views - Multiple ChoiceCalculate the root mean square velocity in m/s of dinitrogen pentoxide gas at 99.5 °C, given that the molar mass of dinitrogen pentoxide is 108 g/mol.495views