7. Gases
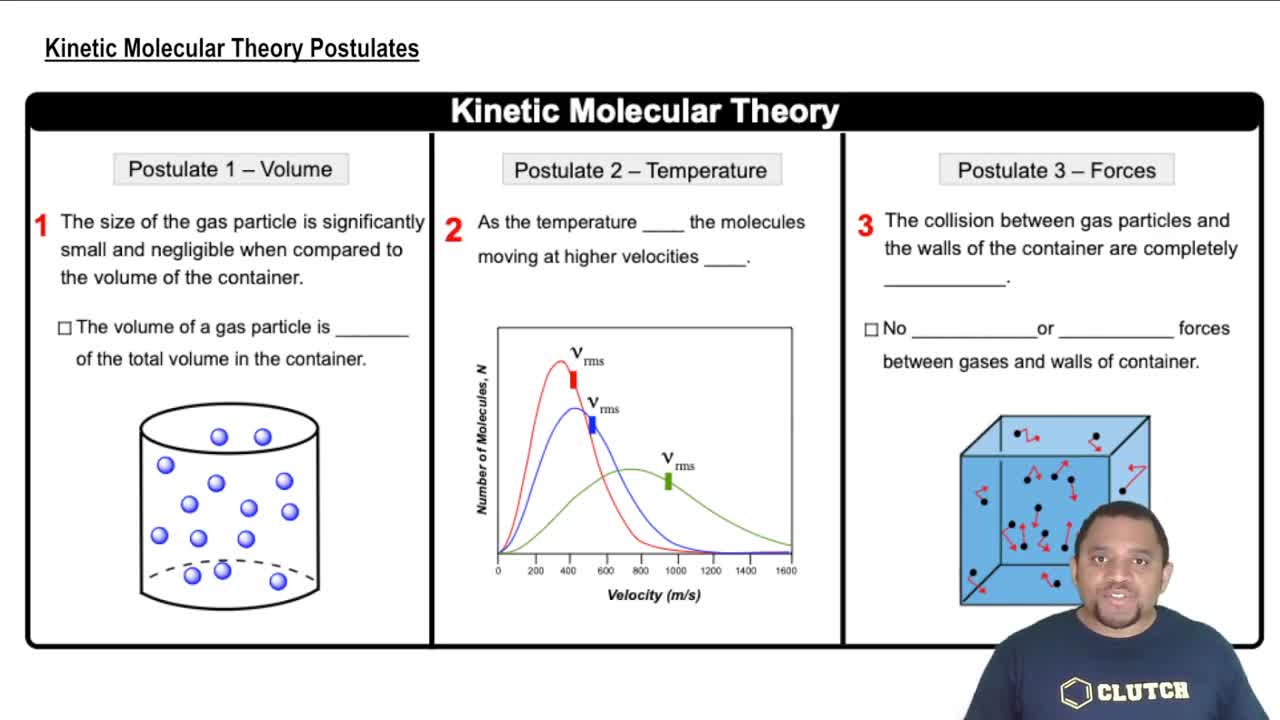
Kinetic Molecular Theory
Practice this topic
- Multiple Choice
Which of the following statements would correctly explain the non-ideal behavior of a gas based on the Kinetic Molecular Theory (KMT)?
a) At high temperatures the attractive forces between molecules becomes negligible.
b) At high pressure the volume of gas molecules become significant.
c) An increase or decrease in the moles of gas causes the gas constant value to change.3174views9rank - Multiple Choice
Which of the following statements is/are true for gas molecules according to the Kinetic Molecular Theory?
I.Increasing the amount of gas molecules increases the pressure by increasing the force of the collisions.
II.Decreasing the temperature of a gas decreases the pressure by increasing the force of the collisions.
III.Decreasing the volume of a gas increases pressure by increasing the frequency of the collisions.4095views9rank - Multiple Choice
Based on the kinetic-molecular theory, which of the following is/are true?
I.At a given temperature, all gases have the same average kinetic energy.
II.At a given temperature, different gases have the same average velocities.
III.The average kinetic energy is proportional to the absolute temperature.3174views13rank2comments - Multiple Choice
Which statement is TRUE about kinetic molecular theory?
a) A single particle does not move in a straight line.
b) The size of the particle is large compared to the volume.
c) The collisions of particles with one another is completely elastic.
d) The average kinetic energy of a particle is not proportional to the temperature.3748views6rank - Textbook Question
At 273 K and 1 atm pressure, 1 mol of an ideal gas occupies 22.4 L. (Section 10.4) (c) In which parts of the atmosphere would you expect gases to behave most ideally (ignoring any photochemical reactions)? [Section 18.1]
991views - Textbook Question
(c) What happens to a gas if you put it under extremely high pressure?
913views - Textbook QuestionIdentify the true statement about deviations from ideal gas behavior. (LO 10.12) (a) The attractive forces between gas particles cause the true volume of the sample to be larger than predicted by the ideal gas law. (b) The attractive forces between gas particles most influence the volume of a sample at low pressure. (c) The volume of the gas particles themselves most influences the volume of the sample at low pressure. (d) The volume of the gas particles themselves causes the true volume of the sample to be larger than predicted by the ideal gas law.710views
- Textbook Question
Which of the following statements is false? (a) Gases are far less dense than liquids. (b) Gases are far more compressible than liquids. (c) Because liquid water and liquid carbon tetrachloride do not mix, neither do their vapors. (d) The volume occupied by a gas is determined by the volume of its container.
1078views1comments