7. Gases
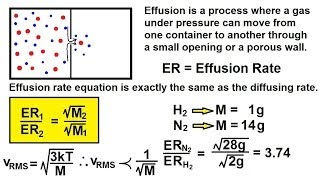
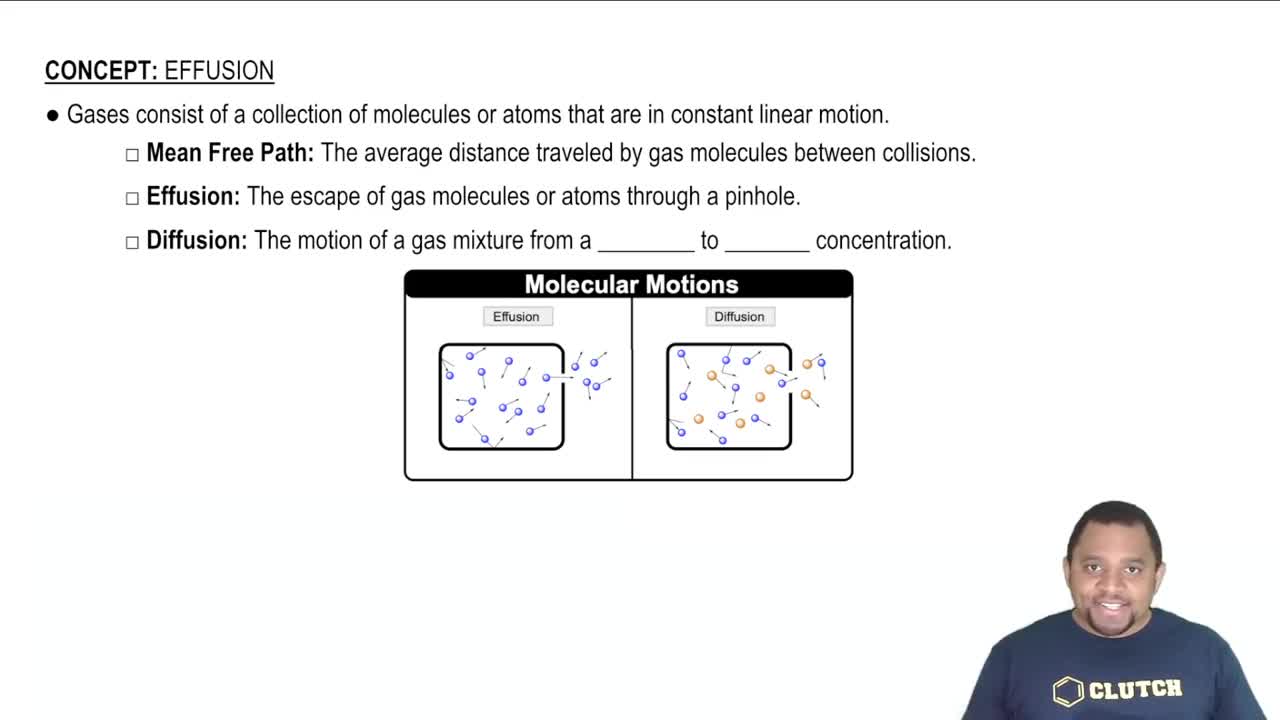
Effusion
Practice this topic
- Multiple Choice
If H2 has an effusion rate that is 3.72 times faster than a gas, what is the identity of the unknown gas?
1526views2rank - Multiple Choice
How many times faster will H2 gas pass through a pinhole into an area of vacuum than O2 gas?
1428views - Multiple Choice
It takes 6.3 minutes for 2.3 L argon to effuse through a semipermeable membrane. How long would it take for 2.3 L of chlorine gas to effuse under similar conditions?
1393views1rank6comments - Multiple ChoiceDetermine the identity of an unknown gas that effuses at a rate 1.48 times as fast as chlorine gas.1240views
- Textbook Question
The apparatus shown here has two gas-filled containers and one empty container, all attached to a hollow horizontal tube. When the valves are opened and the gases are allowed to mix at constant temperature, what is the distribution of atoms in each container?
466views - Textbook Question
A thin glass tube 1 m long is filled with Ar gas at 101.3 kPa, and the ends are stoppered with cotton plugs as shown below. HCl gas is introduced at one end of the tube, and simultaneously NH3 gas is introduced at the other end. When the two gases diffuse through the cotton plugs down the tube and meet, a white ring appears due to the formation of NH4Cl1s2. At which location—a, b, or c—do you expect the ring to form?
685views1rank - Textbook QuestionAn unknown gas is found to diffuse through a porous membrane 2.92 times more slowly than H2. What is the molecular weight of the gas? (a) 17.0 g/mol (b) 5.84 g/mol (c) 8.52 g/mol803views
- Textbook Question
Which one or more of the following statements are true? (a) O2 will effuse faster than Cl2. (b) Effusion and diffusion are different names for the same process. (c) Perfume molecules travel to your nose by the process of effusion. (d) The higher the density of a gas, the shorter the mean free path.
1028views