6. Chemical Quantities & Aqueous Reactions
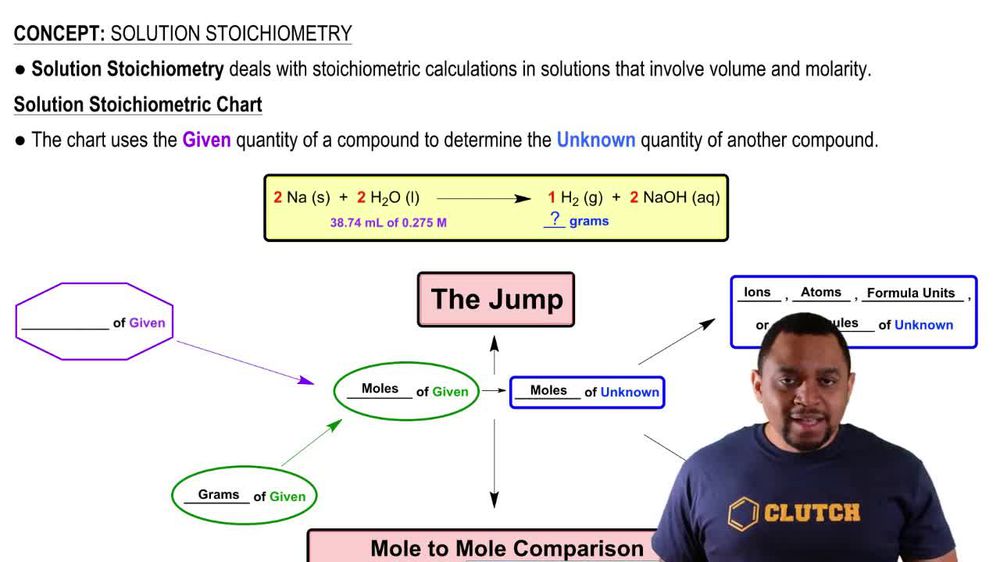
Solution Stoichiometry
6. Chemical Quantities & Aqueous Reactions
Solution Stoichiometry
Showing 9 of 9 videos
Practice this topic
- Multiple Choice
How many milliliters of 0.325 M HCl are needed to react with 16.2 g of magnesium metal?
2 HCl (aq) + Mg (s) → MgCl2 + H2 (g)
4237views7rank4comments - Multiple Choice
What is the molar concentration of a hydrobromic acid solution if it takes 34.12 mL of HBr to completely neutralize 82.56 mL of 0.156 M Ca(OH)2?
2 HBr (aq) + Ca(OH)2 (aq) → CaBr2 (aq) + 2 H2O (l)
3349views7rank2comments - Multiple Choice
Consider the following balanced chemical equation:
H2O+ 2 MnO4– + 3 SO32- → 2 MnO2 + 3 SO42-+ 2 OH–
How many grams of MnO2 (MW:86.94 g/mol) will be created when 25.0 mL of 0.120 M MnO4– (MW:118.90 g/mol) reacts with 32.0 mL of 0.140 M SO32- (MW:80.07 g/mol).
2931views7rank - Textbook QuestionWhen 75.0 mL of a 0.100 M lead(II) nitrate solution is mixed with 100.0 mL of a 0.190 M potassium iodide solu-tion, a yellow-orange precipitate of lead(II) iodide is formed. What is the mass in grams of lead(II) iodide formed? Assume the reaction goes to completion. (LO 4.11, 4.15)(a) 1.729 g(b) 3.458 g(c) 4.380 g (d) 8.760 g1393views1comments
- Textbook QuestionWhat volume of 0.250 M HCl is needed to react completely with 25.00 mL of 0.375 M Na2CO3? (LO 4.15) (a) 75.0 mL(b) 18.8 mL(c) 37.5 mL(d) 33.3 mL1023views
- Textbook QuestionSuccinic acid, an intermediate in the metabolism of food molecules, has a molecular weight of 118.1. When 1.926 g of succinic acid was dissolved in water and titrated, 65.20 mL of 0.5000 M NaOH solution was required to neutralize the acid. How many acidic hydrogens are there in a mol-ecule of succinic acid? (LO 4.16)(a) 1(b) 2 (c) 3 (d) 41095views
- Textbook QuestionThe concentration of a solution of potassium permanganate, KMnO4, can be determined by titration with a known amount of oxalic acid, H2C2O4, according to the following equation: What is the concentration of a KMnO4 solution if 22.35 mL reacts with 0.5170 g of oxalic acid? (LO 4.22) (a) 0.6423 M(b) 0.1028 M(c) 0.4161 M (d) 0.2569 M1900views
- Multiple Choice100.0 mL of 0.100 M sodium phosphate is mixed with 100.0 mL of 0.120 M calcium chloride. What mass of calcium phosphate precipitate is formed?385views