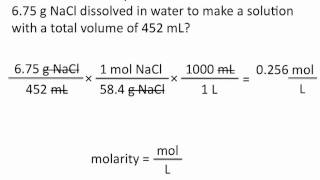
6. Chemical Quantities & Aqueous Reactions
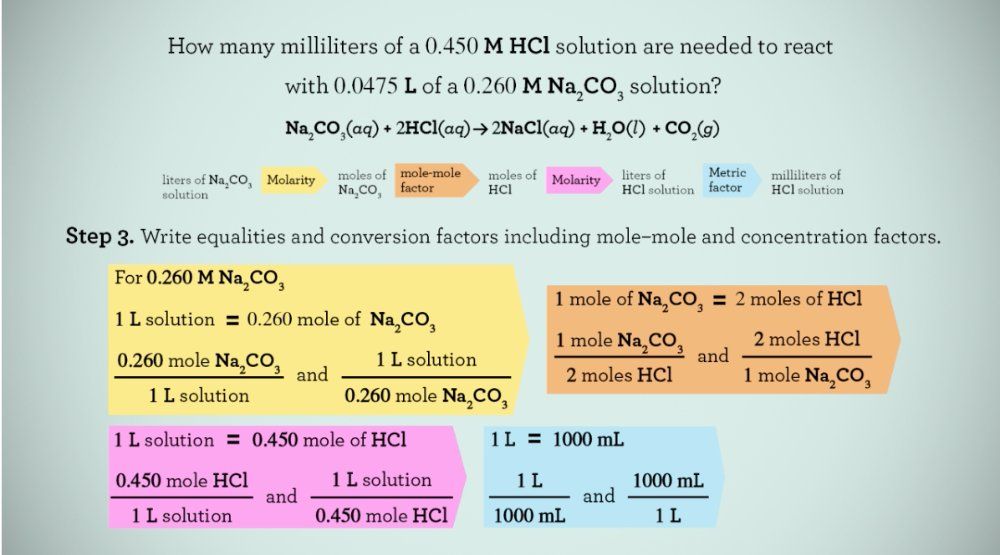
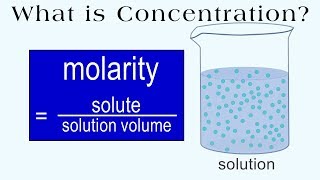
Molarity
Practice this topic
- Multiple Choice
What volume in (µL) of 0.125 M HBr contains 0.170 moles HBr?
3664views21rank1comments - Multiple Choice
Hypernatremia is a medical condition where a patient has high levels of sodium in their blood, and is the result of the body containing too little water. A patient has a measured sodium level of 165 mM. If 30.0 mL of their blood were drawn, what mass (in ng) of sodium would be present?
3507views11rank5comments - Multiple Choice
2.64 grams of an unknown compound was dissolved in water to yield 150 mL of solution. The concentration of the solution was 0.075 M. What was the molecular weight of the substance?
3545views7rank2comments - Multiple Choice
A solution with a final volume of 750.0 mL was prepared by dissolving 30.00 mL of benzene (C6H6, density = 0.8787 g/mL) in dichloromethane. Calculate the molarity of benzene in the solution.
4342views4rank4comments - Textbook Question
What is the molarity of a solution prepared by dissolving 10.19 g of ethanol (CH3CH2OH) in enough water to produce 250.0 mL of solution? (LO 4.1) (a) 0.8848 M (b) 18.08 M (c) 1.130 M (d) 0.01808 M
1038views1rank - Textbook Question
What is the molarity of Na+ in a solution of NaCl whose salinity is 5.6 if the solution has a density of 1.03 g>mL?
574views - Textbook Question
What is the molarity of each of the following solutions: (b) 5.25 g of Mn(NO3)2⋅2H2O in 175 mL of solution,
589views - Textbook Question
How many moles of solute are present in each of the following solutions? (b) 175 mL of 0.67 M glucose (C6H12O6)
893views