6. Chemical Quantities & Aqueous Reactions
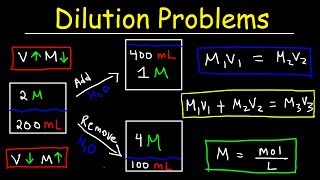
Dilutions
6. Chemical Quantities & Aqueous Reactions
Dilutions
Additional 2 creators.
Learn with other creators
Showing 5 of 5 videos
Practice this topic
- Multiple Choice
To what final volume would 100 mL of 5.0 M KCl have to be diluted in order to make a solution that is 0.54 M KCl?
4955views8rank1comments - Multiple Choice
If 880 mL of water is added to 125.0 mL of a 0.770 M HBrO4 solution what is the resulting molarity?
4856views14rank1comments - Open Question
What volume of 12.0 M HCl is required to make 75.0 mL of 3.50 M HCl?
934views - Open QuestionA standard solution is prepared for the analysis of fluoxymesterone1745views
- Open Question
How many ml of a 3 mol/L NaOH solution are required to make 100 ml of a 0.2 mol/L NaOH solution?
754views - Open Question
Calculate the molarity of a solution of FD&C blue dye no. 1
717views - Multiple ChoiceA 100.0 mL sample is prepared from a 250.0 mL stock solution by pipetting 5.00 mL of the stock solution and diluting it until it had a molarity of 2.5 * 10^-4 M. What is the molarity of the original stock solution?577views
- Multiple ChoiceA flask contains 50.0 mL of 1.00 M sodium hydroxide. Its contents are fully transferred to a larger flask, and enough water is added to prepare 1.00 L of solution. What is the concentration of sodium hydroxide in the diluted solution?493views