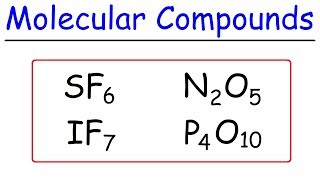
3. Chemical Reactions
Naming Molecular Compounds
3. Chemical Reactions
Naming Molecular Compounds
Additional 5 creators.
Learn with other creators
Showing 8 of 8 videos
Practice this topic
- Multiple Choice
Give the systematic name for the following compound:SeF6
5752views19rank - Multiple Choice
Give the systematic name for the following compound:IO5
5073views16rank - Multiple Choice
Give the systematic name for the following compound:N2S4
5443views17rank1comments - Multiple ChoiceConsider the following representation of a chemical change.
2 KClO3(s) → 2 KCl (s)+ 3 O2(g)
Which of the following correctly describes this process?1246views - Open Question
What is the name of the compound P4O10?
1883views - Open Question
What is the name of N2Cl4? Explain how you determined the bond type and the steps you used to determine the naming convention for the compound.
1858views - Open Question
The chemical name for table salt is sodium chloride, or simply NaCl. What type of chemical is NaCl?
2291views - Open Question
The formula for laughing gas is N2O. What is the IUPAC name for N2O?
1483views