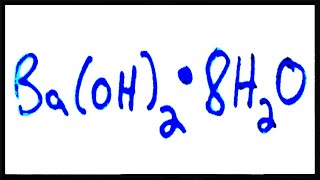3. Chemical Reactions
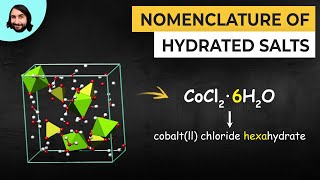
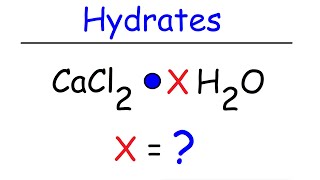
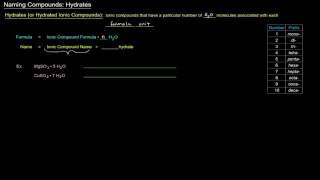
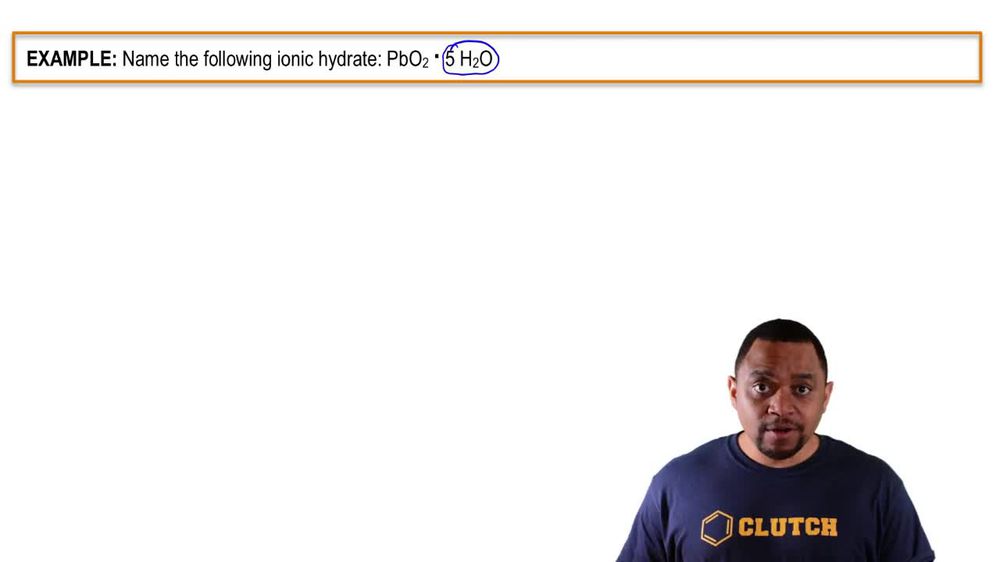
Naming Ionic Hydrates
Learn with other creators
Practice this topic
- Multiple Choice
Give the systematic name from the given formula:K2Cr2O7 ∙ 3 H2O
3880views16rank1comments - Multiple Choice
Write the chemical formula for the following compound:Calcium selenide hexahydrate
3554views11rank - Textbook Question
Write the name from the formula or the formula from the name for each hydrated ionic compound.
a. CoSO4•7 H2O
b. iridium(III) bromide tetrahydrate
c. Mg(BrO3)2•6 H2O
d. potassium carbonate dihydrate
979views - Textbook Question
Write the name from the formula or the formula from the name for each hydrated ionic compound.
a. cobalt(II) phosphate octahydrate
b. BeCl2•2 H2O
c. chromium(III) phosphate trihydrate
d. LiNO2•H2O
618views - Textbook Question
Washing soda, a compound used to prepare hard water for washing laundry, is a hydrate, which means that a certain number of water molecules are included in the solid structure. Its formula can be written as Na2CO3 # xH2O, where x is the number of moles of H2O per mole of Na2CO3. When a 2.558-g sample of washing soda is heated at 125 C, all the water of hydration is lost, leaving 0.948 g of Na2CO3. What is the value of x?
2993views1comments - Textbook Question
Epsom salts, a strong laxative used in veterinary medicine, is a hydrate, which means that a certain number of water molecules are included in the solid structure. The formula for Epsom salts can be written as MgSO4 # xH2O, where x indicates the number of moles of H2O per mole of MgSO4. When 5.061 g of this hydrate is heated to 250 C, all the water of hydration is lost, leaving 2.472 g of MgSO4. What is the value of x?
1350views - Multiple ChoiceWhich of the following is the correct chemical formula for barium hydroxide octahydrate?91views
- Multiple ChoiceWhich of the following is the correct chemical formula for cobalt(II) chloride hexahydrate?67views