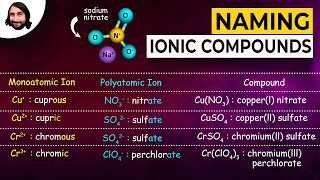
3. Chemical Reactions
Naming Ionic Compounds
3. Chemical Reactions
Naming Ionic Compounds
Additional 4 creators.
Learn with other creators
Showing 7 of 7 videos
Practice this topic
- Multiple Choice
Provide the name for the following compound:Al(BrO4)3
7267views37rank1comments - Multiple Choice
Provide the name for the following compound:CoCO3
7393views37rank - Open Question
Determine the name for TiCO3. Remember that titanium forms several ions.
1292views - Open Question
Write the name for Sn(SO4)2. Remember that Sn forms several ions.
1228views - Open Question
Name Ag3AsO4 by analogy to the corresponding compound containing phosphorus in place of arsenic.
1110views1rank - Open Question
In a compound that contains monatomic ions, which of the following gets named first?
1344views - Multiple ChoiceWhat is the chemical formula for the compound formed between beryllium and the chromate ion?1013views
- Multiple ChoiceWhat is the correct formula for the ionic compound formed between P^3- and Ca^2+?822views