3. Chemical Reactions
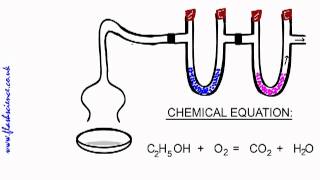
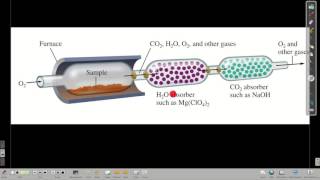
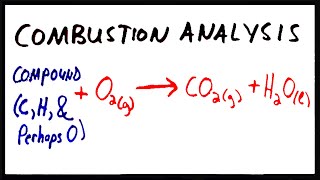
Combustion Analysis
Practice this topic
- Open Question
The combustion of 4.16 grams of a compound which contains only C,H,O and F yields 7.7 g CO2 and 2.52 g H2O. Another sample of the compound with a mass of 3.63 g is found to contain 0.58 g F. What is the empirical formula of the compound?
2259views25rank4comments - Multiple ChoiceWhen the following equation is balanced
C5H10(l) + O2(g) → CO2(g) + H2O(g)
What is the coefficient for carbon dioxide?1309views - Multiple Choice
A compound composed of carbon, hydrogen and nitrogen undergoes a combustion reaction to produce 264.21 g CO2, 63.06 g H2O and 46.4 g NO2. Determine its empirical formula.
1624views11rank - Multiple Choice
An unknown compound is composed of carbon, hydrogen and oxygen. A 4.30 g sample is ignited and creates 8.59 g CO2 and 3.52 g H2O. If the molar mass is 176.22 g/mol, what is the molecular formula?
1539views12rank - Multiple Choice
In the presence of a small amount of oxygen a combustion reaction will not only produce carbon dioxide, but also carbon monoxide. The incomplete combustion of naphthalene, a hydrocarbon used in many dyes, produced 2.80 g CO, 4.40 g CO2 and 1.44 g H2O. Determine its empirical formula.
1025views9rank1comments - Textbook Question
Combustion analysis is performed on 0.50 g of a hydrocar-bon and 1.55 g of CO2, and 0.697 g of H2O are produced. The mass spectrum for the hydrocarbon is provided below. What is the molecular formula? (LO 3.12 and 3.13)
(a) C5H11 (b) C8H18 (c) C11H10 (d) C10H22
612views - Textbook Question
(b) What products form in this reaction?
446views - Textbook Question
(a) When a compound containing C, H, and O is completely combusted in air, what reactant besides the hydrocarbon is involved in the reaction?
1254views