15. Chemical Kinetics
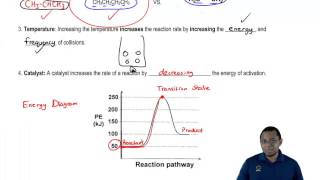
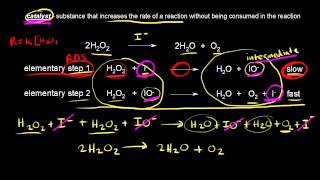
Catalyst
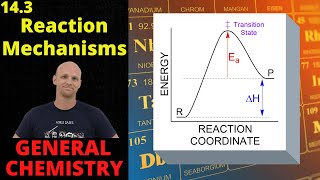
15. Chemical Kinetics
Catalyst
Additional 5 creators.
Learn with other creators
Showing 8 of 8 videos
Practice this topic
- Multiple Choice
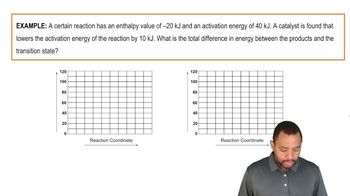
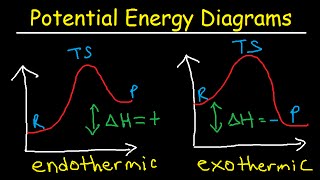
Which of the following statements is true regarding the energy diagram provided?
i. The reaction is endothermic.
ii. Activation energy would be less than + 10 kJ after a catalyst is added.
iii. The reaction absorbs energy.
iv. Activation energy would be greater than + 10 kJ after a catalyst is added.
2341views18rank - Multiple ChoiceWhich of the following is true about a catalyst?1655views1rank
- Open QuestionHow are the following aspects of a reaction affected by the addition of a catalyst?1575views
- Open Question
A substance that influences the reaction but does not participate in the reaction is a
1129views - Open Question
Enzymes are catalysts because they lower the ______________ of a chemical reaction.
1072views1rank - Open Question
A(n) ___ is a substance that speeds up a chemical reaction without being used up in the reaction.
883views - Multiple ChoiceAt 300 K, a catalyst lowers the activation barrier from 20 kJ/mol to 10 kJ/mol. Which of the following statements is true regarding the rate constant for the catalyzed reaction compared to the uncatalyzed reaction? (R = 8.314 J mol⁻¹ K⁻¹)865views
- Multiple ChoiceWhy are enzymes referred to as catalysts in biochemical reactions?767views