7. Gases
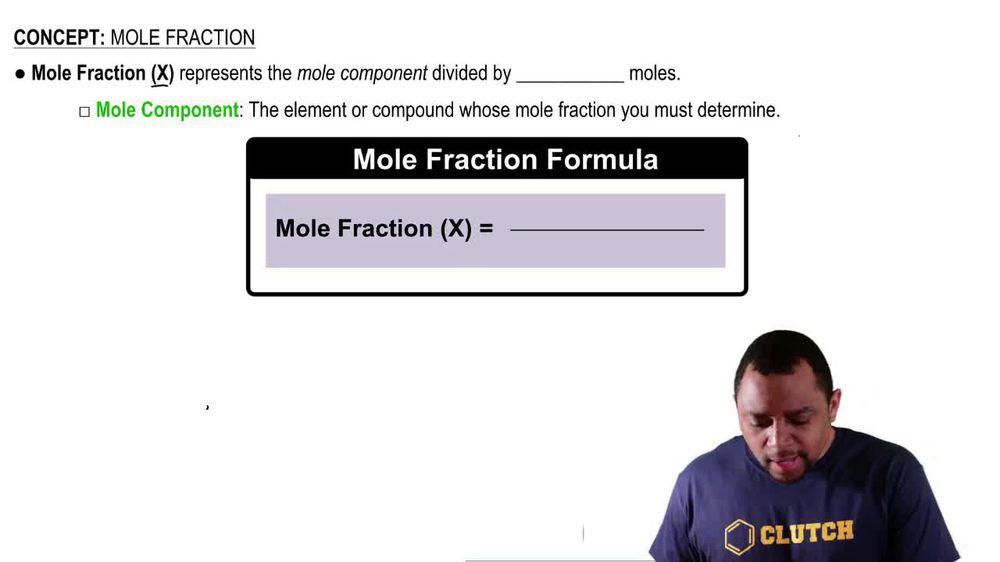
Mole Fraction of Gases
7. Gases
Mole Fraction of Gases
Showing 8 of 8 videos
Practice this topic
- Multiple Choice
A reaction vessel is composed of 20.3 g Cl2, 4.27 g N2 and 10.8 g Ne. Calculate the mole fraction of nitrogen.
1455views6rank - Textbook QuestionCalculate the average molecular weight of air from the datagiven in Table 10.1.864views
- Textbook QuestionDuring a person's typical breathing cycle, the CO2 concentration in the expired air rises to a peak of 4.6% by volume.(a) Calculate the partial pressure of the CO2 in the expiredair at its peak, assuming 1 atm pressure and a body temperature of 37 °C.1363views
- Textbook Question
The atmospheric concentration of CO2 gas is presently 407 ppm (parts per million, by volume; that is, 407 L of every 106 L of the atmosphere are CO2). What is the mole fraction of CO2 in the atmosphere?.
1951views - Textbook Question
What is the mole fraction of oxygen gas in air (see Table 5.3)? What volume of air contains 10.0 g of oxygen gas at 273 K and 1.00 atm?
2666views1rank - Multiple ChoiceWhich of the following is the approximate mole fraction of argon (Ar) in Earth's atmosphere?64views