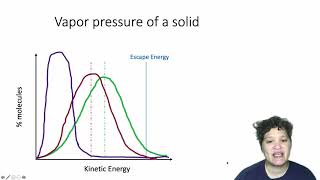
13. Liquids, Solids & Intermolecular Forces
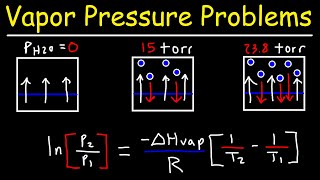
Clausius-Clapeyron Equation
13. Liquids, Solids & Intermolecular Forces
Clausius-Clapeyron Equation
Showing 11 of 11 videos
Practice this topic
- Multiple Choice
Vapor pressure measurements at various temperature values are given below. Determine the molar heat of vaporization for cyclohexane.
1450views11rank1comments - Multiple Choice
Benzene has a heat of vaporization of 30.72 kJ/mol and a normal boiling point of 80.1°C. At what temperature does benzene boil when the external pressure is 405 torr?
5987views6rank4comments - Textbook QuestionWhat is the vapor pressure of SiCl4 in mm Hg at 30.0 °C? (See Problem 11.27.)723views
- Textbook QuestionDichloromethane, CH2Cl2, is an organic solvent used for removing caffeine from coffee beans. The following table gives the vapor pressure of dichloromethane at various tem- peratures. Fill in the rest of the table, and use the data to plot curves of Pvap versus T and ln Pvap versus 1/T.701views
- Textbook QuestionChoose any two temperatures and corresponding vapor pressures in the table given in Problem 11.30, and use those values to calculate ΔHvap for dichloromethane in kJ/mol. How does the value you calculated compare to the value you read from your plot in Problem 11.32?676views
- Textbook QuestionDichlorodifluoromethane, CCl2F2, one of the chlorofluo- rocarbon refrigerants responsible for destroying part of the Earth's ozone layer, has Pvap = 40.0 mm Hg at -81.6 °C and Pvap = 400 mm Hg at -43.9 °C. What is the normal boiling point of CCl2F2 in °C?1120views2comments
- Multiple ChoiceA substance has a normal boiling point of 76 °C and an enthalpy of vaporization (ΔvapH) of 38.7 kJ/mol. Using the Clausius-Clapeyron equation, determine the vapor pressure (in mbar) of this substance at 29 °C.577views
- Multiple ChoiceBenzene has a heat of vaporization of 30.72 kJ/mol and a normal boiling point of 80.1 °C. At what temperature does benzene boil when the external pressure is 577 mbar?511views