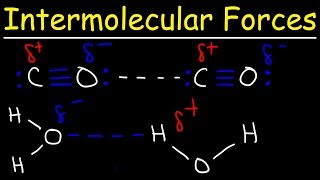
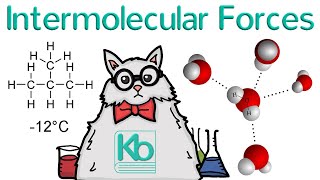
13. Liquids, Solids & Intermolecular Forces
Intermolecular Forces and Physical Properties
13. Liquids, Solids & Intermolecular Forces
Intermolecular Forces and Physical Properties
Showing 10 of 10 videos
Practice this topic
- Multiple Choice
Which of the following will have the lowest boiling point?
3043views9rank1comments - Multiple Choice
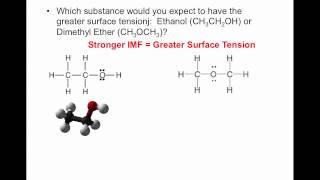
Which molecules would most likely cause a liquid to have the lowest viscosity?
1933views11rank - Multiple ChoiceWhich of the following is a false statement about dispersion forces?1179views
- Multiple ChoiceWhich of the following statements is false about the changes occurring when ice is heated in an open beaker from −10.0℃ to water vapor at 110.0℃?915views
- Textbook QuestionThree identical tubes are filled with different liquids; water, ethylene glycol, and olive oil. A small steel sphere was dropped into the tube, and the time it took to fall to the bottom was recorded.
Which property of liquids is responsible for the differences in time for the sphere to fall through the liquid? (LO 11.1)(a) Surface tension(b) Boiling point(c) Viscosity(d) Vapor pressure
430viewsTextbook QuestionWhich organic compound has the lowest viscosity? (LO 11.2)701viewsTextbook QuestionArrange the following molecules from lowest to highest boilingpoint. (LO 8.9)(I) (II) (III) (IV) (a) I 6 II 6 III 6 IV(b) II 6 III 6 I 6 IV(c) IV 6 II 6 III 6 I(d) IV 6 I 6 II 6 III697viewsTextbook QuestionWhich type of intermolecular force accounts for each of these differences? (a) CH3OH boils at 65 °C; CH3SH boils at 6 °C. (d) Acetone boils at 56 °C, whereas 2-methylpropane boils at -12 °C.
440views