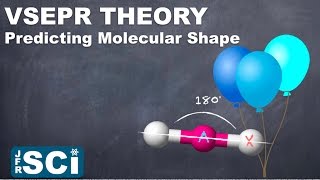
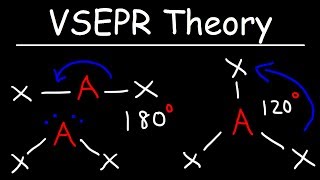
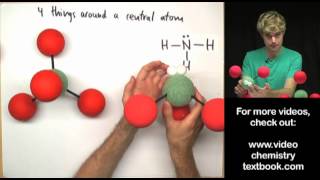
12. Molecular Shapes & Valence Bond Theory
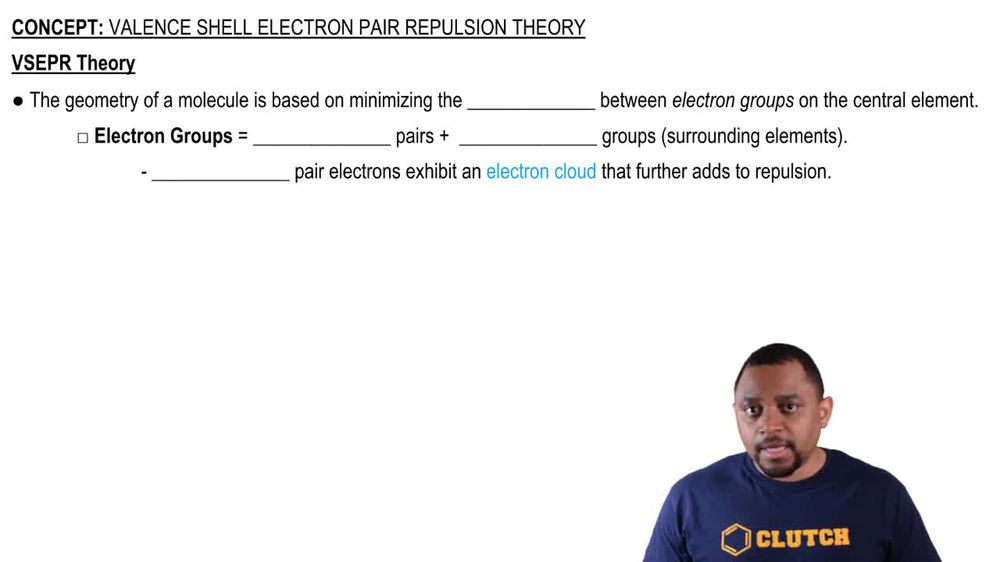
Valence Shell Electron Pair Repulsion Theory
12. Molecular Shapes & Valence Bond Theory
Valence Shell Electron Pair Repulsion Theory
Additional 8 creators.
Learn with other creators
Showing 11 of 11 videos
Practice this topic
- Multiple ChoiceWhich of the following statements is false about VSEPR theory?1144views
- Multiple ChoiceWhat two orbitals are participating in the overlap region that is part of the σ bond between the carbon and hydrogen atoms in acetylene?801views
- Textbook Question
Draw sketches illustrating the overlap between the following orbitals on two atoms: (b) the 2pz orbital on each atom (assume both atoms are on the z-axis) (c) the 2s orbital on one atom and the 2pz orbital on the other atom.
927views - Textbook Question
Draw sketches illustrating the overlap between the following orbitals on two atoms: (a) the 2s orbital on each atom
587views1rank - Textbook QuestionHow would you expect the extent of overlap of the bondingatomic orbitals to vary in the series IF, ICl, IBr, and I2?Explain your answer.724views
- Textbook Question
A series of anions is shown below:
The anion on the far right is called 'BARF' by chemists, as its common abbreviation sounds similar to this word. (a) What is the central atom and the number of electronpair domains around the central atom in each of these anions?
356views - Multiple ChoiceAccording to VSEPR theory, which of the following species has all its atoms lying in the same plane?574views
- Multiple ChoiceAccording to the Valence Shell Electron Pair Repulsion (VSEPR) Theory, how many electron groups are around the central atom in the molecule OF2?548views