1. Intro to General Chemistry
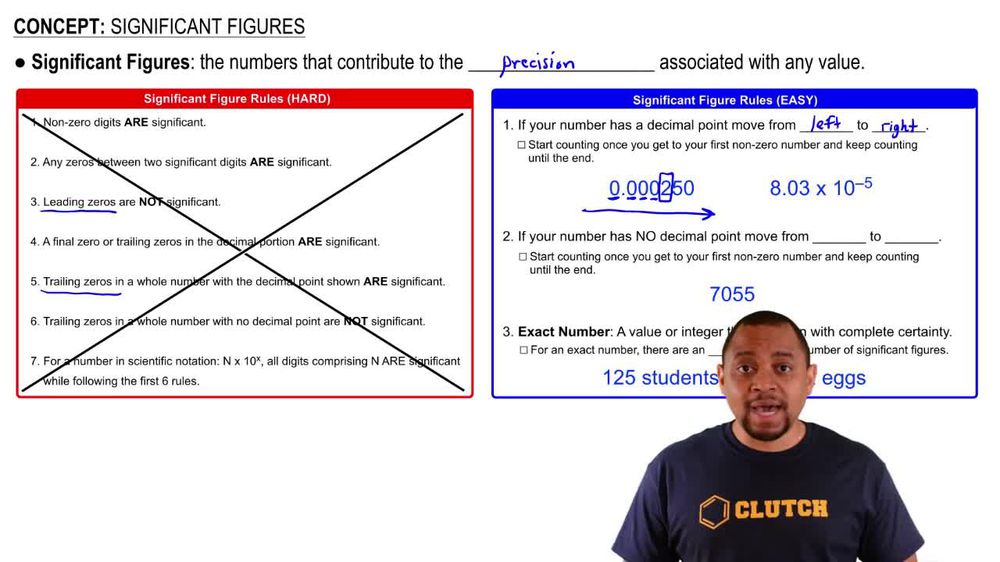
Significant Figures
1. Intro to General Chemistry
Significant Figures
Additional 4 creators.
Learn with other creators
Showing 7 of 7 videos
Practice this topic
- Open Question
How many sig figs does each number contain?
a) 100. min
b) 17.3 x 103 mL
c) 10 apples
4073views53rank4comments - Multiple Choice
Indicate the number of significant figures in the following:
A liter is equivalent to 1.059 qt.
5321views44rank - Multiple Choice
How many significant figures are contained in the following measurement?
3,482,005 mg
2307views32rank1comments - Multiple ChoiceWhich of the following has the largest number of significant figures?1362views
- Multiple ChoiceHow many significant figures should be reported for the following calculation:
5.2110 × 12.20 ÷ (11.44 − 2.113)1379views - Open Question
How many significant figures are in the measurement 603.040 g ?
1076views - Open Question
Indicate the number of significant figures in each of the following
1354views - Open Question
Determine the number of significant figures in each of the following measurements
1024views