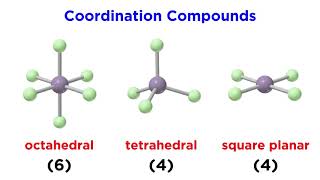
24. Transition Metals and Coordination Compounds
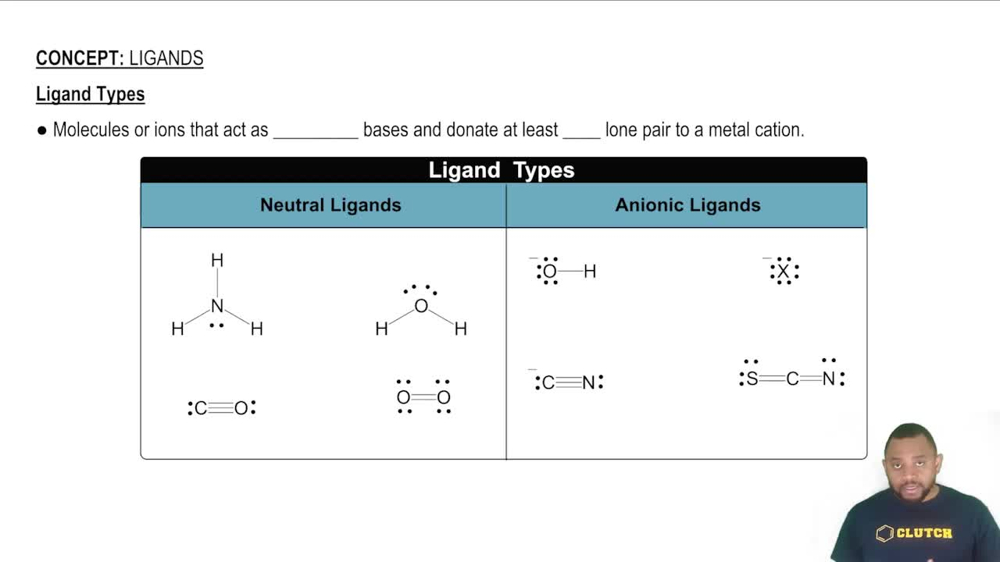
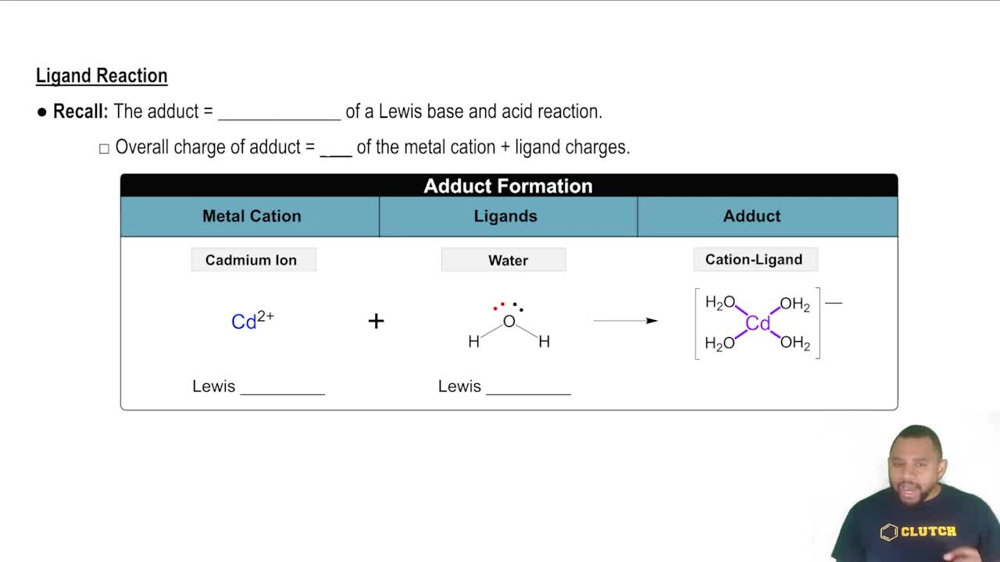
Ligands
24. Transition Metals and Coordination Compounds
Ligands
Additional 7 creators.
Learn with other creators
Showing 10 of 10 videos
Practice this topic
- Open Question
Determine the geometry for the following complex ion:[Cr(NH3)4Cl2]2+
1061views4comments - Multiple ChoiceAn ion that contains a central metal ion bound to one or more ligands is called a ________.965views
- Multiple ChoiceA coordination compound consists of _________.1025views
- Multiple ChoiceEthylenediamine (shown below) would be called a __________.999views
- Multiple ChoiceEDTA is a ligand that can wrap itself completely around the metal ion, donating up to six pairs of electrons; this ligand is a type of __________.1059views1rank
- Textbook Question
What is the oxidation state of the metal in each of the complexes?
a. AgCl2–
b. [Cr(H2O)5Cl]2+
c. [Co(NCS)4]2–
d. [ZrF8]4–
e. [Fe(EDTA)(H2O)]–
89views - Textbook Question
What is the oxidation state of the metal in each of the complexes?
a. [Ni(CN)5]3–
b. Ni(CO)4
c. [Co(en)2(H2O)Br]2+
d. [Cu(H2O)2(C2O4)2]2–
e. Co(NH3)3(NO2)3
71views - Textbook Question
Identify the oxidation state of the metal in each of the following compounds.
(a) Co(NH3)3(NO2)3
(b) [Ag(NH3)2]NO3
(c) K3[Cr(C2O4)2Cl2]
(d) Cs[CuCl2]
90views