24. Transition Metals and Coordination Compounds
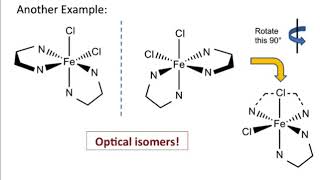
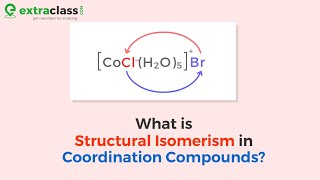
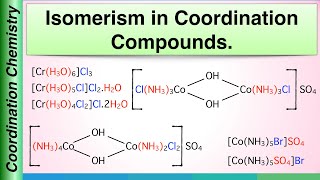
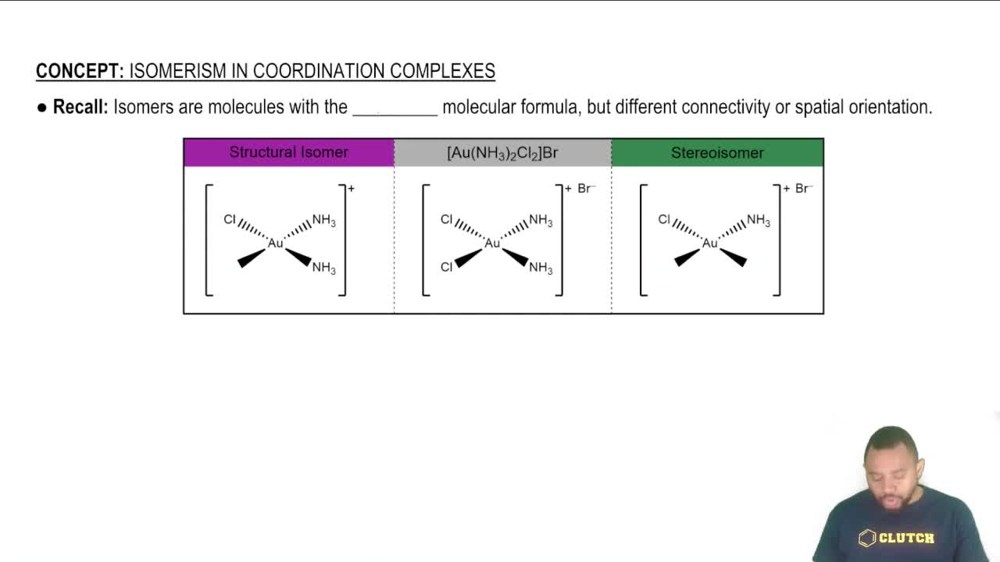
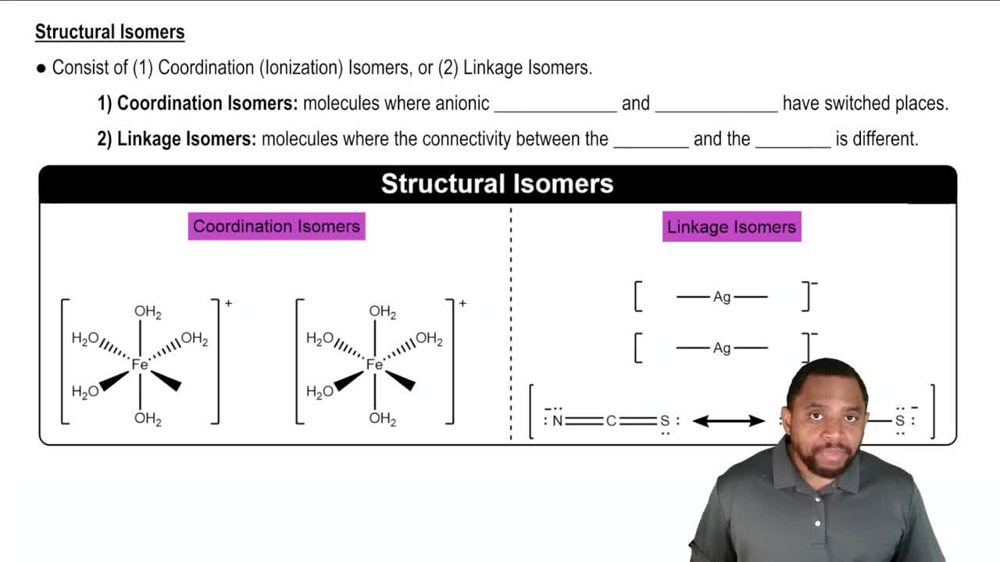
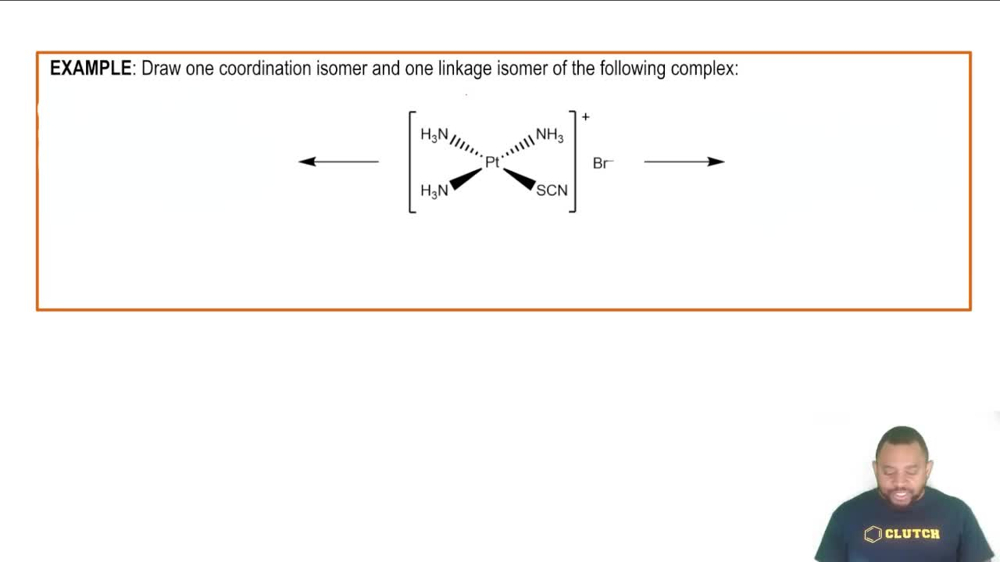
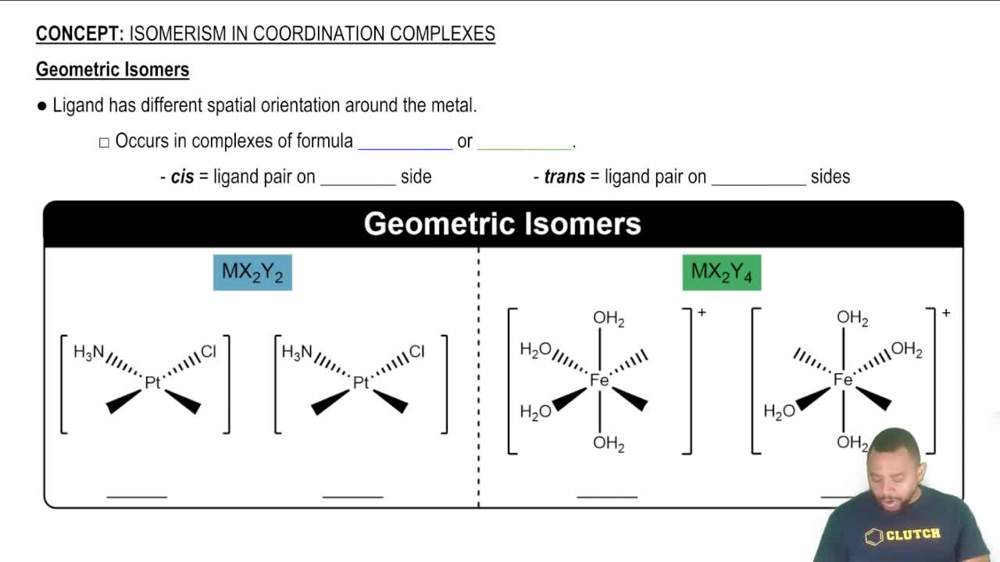
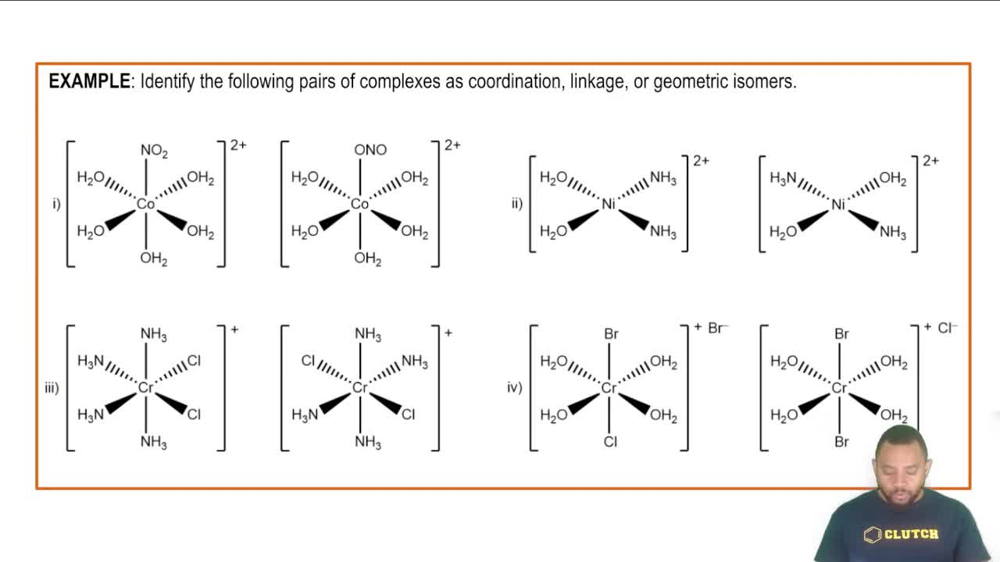
Isomerism in Coordination Complexes
Learn with other creators
Practice this topic
- Multiple ChoiceWhich of the following is true for coordination isomers?1096views
- Multiple ChoiceHow many stereoisomers are possible for Ni(NH3)2Br2?1165views
- Multiple ChoiceWrite the formula for the coordination compound that is named as ammonium tetrachlorocuprate(II).1181views2rank
- Multiple Choice
Which of the following complexes cannot have geometric isomers?
i) [PtCl2(NH3)2]
ii) K4[Fe(CN)4(OH)2]
iii) [Ag(NH3)2]Cl
iv) [Ni(H2O)2(NH3)2]Br2
927views3rank - Textbook Question
In acidic aqueous solution, the complex trans-[Co(en)2Cl1]2+(aq) undergoes the following substitution reaction:
trans-[Co(en)1Cl2]+(aq) + H2O(l) → trans-[Co(en)2(H2O)Cl]2+(aq) + Cl–(aq)
The reaction is first order in trans-[Co(en)2Cl2]+(aq), and the rate constant at 25°C is 3.2×10–5 s–1.
(d) Is the reaction product chiral or achiral? Explain.
76views - Textbook Question
Constitutional isomers of a ruthenium(II) coordination compound are shown below.
(a) Give the formula and name for structures 1-3.
(b) Which structures are linkage isomers?
(c) Which structures are ionization isomers?
93views - Textbook Question
Six isomers for a square planar palladium(II) complex that contains two Cl-and two SCN-ligands are shown below.
(a) Which structures are cis-trans isomers?
(b) Which structures are linkage isomers?
112views - Textbook Question
Tell how many diastereoisomers are possible for each of the following complexes, and draw their structures.
(a) Pt(NH3)3Cl (square planar)
(b) [FeBr2Cl2(en)]-
102views