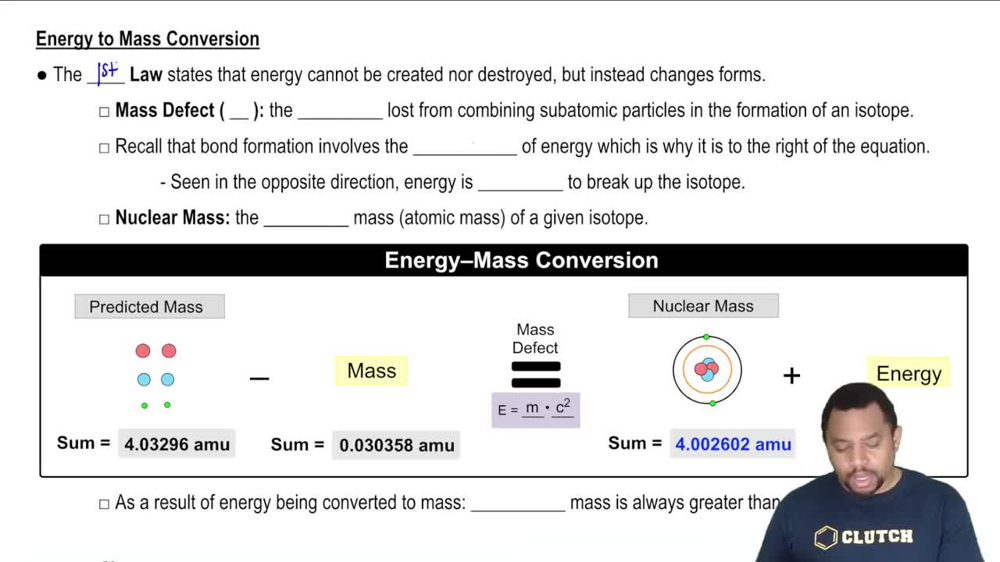
21. Nuclear Chemistry
Mass Defect
Practice this topic
- Multiple Choice
Calculate the mass defect (in mg) for the following isotope. (1 neutron = 1.00866 amu, 1 proton = 1.00727 amu, & 1 electron = 0.00055 amu).
14C
786views1rank1comments - Textbook Question
Calculate the quantity of energy produced per mole of U-235 (atomic mass = 235.043922 amu) for the neutron-induced fission of U-235 to produce Te-137 (atomic mass = 136.9253 amu) and Zr-97 (atomic mass = 96.910950 amu) (discussed in Problem 58).
107views - Textbook Question
Complete each nuclear equation and calculate the energy change (in J/mol of reactant) associated with each (Al-27 = 26.981538 amu, Am-241 = 241.056822 amu, He-4 = 4.002603 amu, Np-237 = 237.048166 amu, P-30 = 29.981801 amu, S-32 = 31.972071 amu, and Si-29 = 28.976495 amu).
a. 2713Al + 42He → 3015P + ____
110views - Textbook Question
The thermite reaction, Fe2O31s2 + 2 Al1s2 ¡2 Fe1s2 + Al2O31s2, H = -851.5 kJ>mol, is one of the most exothermic reactions known. Because the heat released is sufficient to melt the iron product, the reaction is used to weld metal under the ocean. How much heat is released per mole of Al2O3 produced? How does this amount of thermal energy compare with the energy released when 2 mol of protons and 2 mol of neutrons combine to form 1 mol of alpha particles?
78views - Textbook Question
The atomic masses of hydrogen-2 (deuterium), helium-4, and lithium-6 are 2.014102 amu, 4.002602 amu, and 6.0151228 amu, respectively. For each isotope, calculate
(c) the nuclear binding energy per nucleon.
95views - Multiple ChoiceHelium-3, an electron, a neutron, and a proton have masses of 3.016029 amu, 5.486 × 10^-4 amu, 1.00866 amu, and 1.00728 amu, respectively. What is the mass defect for the formation of helium-3 in amu?559views
- Multiple ChoiceFor an atom of oxygen-18 (18 8O), the mass defect can be converted to an equivalent binding energy (E_b) using Einstein's equation. Which of the following expressions correctly represents the binding energy of the nucleus?71views