20. Electrochemistry
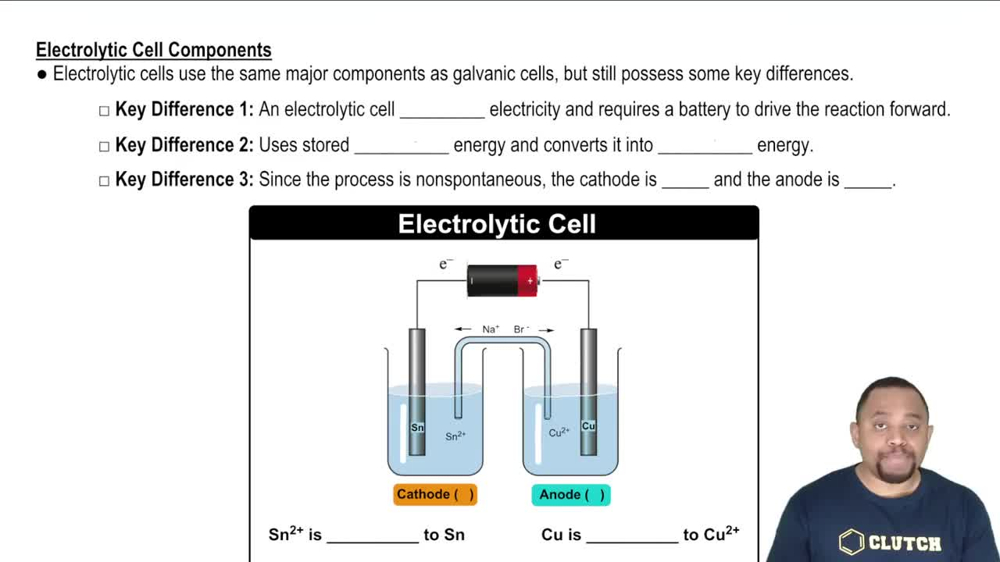
Electrolytic Cell
Practice this topic
- Multiple Choice
If the overall redox reaction for an electrolytic cell is given below, what will happen to the mass of the cobalt electrode?
Co2+ (aq) + Cu (s) ⇌ Co (s) + Cu2+(aq)
1188views1rank - Textbook Question
Magnesium, the element, is produced commercially by electrolysis from a molten salt (the 'electrolyte') using a cell similar to the one shown here. (c) Recall that in an electrolytic cell the anode is given the + sign and the cathode is given the – sign, which is the opposite of what we see in batteries. What half-reaction occurs at the anode in this electrolytic cell?
850views - Textbook QuestionSketch a cell with inert electrodes suitable for electrolysis of aqueous CuBr2. (b) Indicate the direction of electron and ion flow.658views
- Textbook Question
Porous pellets of TiO2 can be reduced to titanium metal at the cathode of an electrochemical cell containing molten CaCl2 as the electrolyte. When the TiO2 is reduced, the O2-ions dis-solve in the CaCl2 and are subsequently oxidized to O2 gas at the anode. This approach may be the basis for a less expensive process than the one currently used for producing titanium.
(c) Write balanced equations for the anode, cathode, and overall cell reactions.
616views - Textbook Question
Porous pellets of TiO2 can be reduced to titanium metal at the cathode of an electrochemical cell containing molten CaCl2 as the electrolyte. When the TiO2 is reduced, the O2-ions dis-solve in the CaCl2 and are subsequently oxidized to O2 gas at the anode. This approach may be the basis for a less expensive process than the one currently used for producing titanium.
(a) Label the anode and cathode, and indicate the signs of the electrodes.
364views - Multiple ChoiceHow many faradays of electricity are required to liberate 9g of aluminum in an electrolytic cell, given that the molar mass of aluminum is 27 g/mol?689views
- Multiple ChoiceIn an electrolytic cell, what is the role of the electrical charge?746views
- Multiple ChoiceIn an electrolytic cell, where does oxidation occur?731views