18. Aqueous Equilibrium
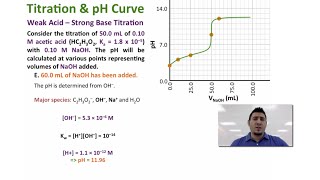
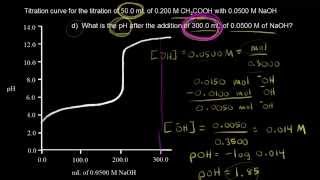
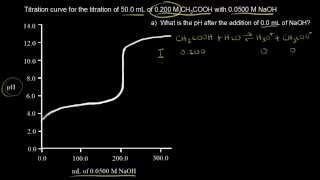
Titrations: Weak Acid-Strong Base
18. Aqueous Equilibrium
Titrations: Weak Acid-Strong Base
Showing 6 of 6 videos
Additional 3 creators.
Learn with other creators
Showing 6 of 6 videos
Practice this topic
- Multiple ChoiceCalculate the volume (mL) of 0.500 M NaOH required to reach the equivalence point in the titration of 25.0 mL of 0.650 M HF. Ka for HF = 3.5 × 10−4.1162views
- Multiple ChoiceFor a titration of a weak acid with a strong base, the pH at the equivalence point ________.1065views
- Multiple Choice20.0 mL of 0.500 M HC2H3O2 is titrated with 0.350 M KOH. Determine the pH after 15.0 mL of the base has been added. Ka for HC2H3O2 = 1.8 × 10−5.838views
- Multiple Choice20.0 mL of 0.500 M HC2H3O2 is titrated with 0.350 M KOH. Determine the pH after 15.0 mL of the base has been added. Ka for HC2H3O2 = 1.8 × 10−5.927views
- Open Question
How will you detect the endpoint of the titration of the borate ion with HCl?
508views - Open Question
Calculate the pH when 50.0 mL of 0.150 M KOH is mixed with 20.0 mL of 0.300 M HBrO (Ka = 2.5 × 10-9)
514views