18. Aqueous Equilibrium
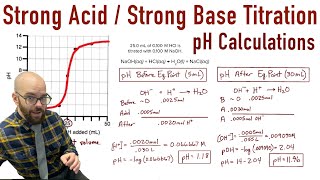
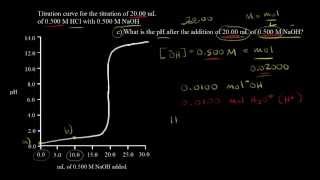
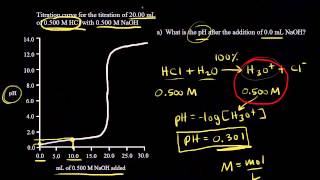
Titrations: Strong Acid-Strong Base
Learn with other creators
Practice this topic
- Multiple Choice
Calculate the pH of the solution resulting from the mixing of 175.0 mL of 0.250 M HNO3 with 75.0 mL of 0.200M Ba(OH)2.
2034views1rank4comments - Multiple Choice
Calculate the pH of the solution resulting from the mixing of 175.0 mL of 0.250 M HNO3 with 75.0 mL of 0.200 M Ba(OH)2.
836views5rank1comments - Multiple Choice
Calculate the pH of the solution resulting from the titration of 110.0 mL of 0.300 M HCl with 330.0 mL of 0.100 M LiOH.
876views4rank - Textbook Question
A 20.0-mL sample of 0.200 M HBr solution is titrated with 0.200 M NaOH solution. Calculate the pH of the solution after the following volumes of base have been added:
(c) 20.0 mL.
118views - Textbook Question
A 20.0-mL sample of 0.200 M HBr solution is titrated with 0.200 M NaOH solution. Calculate the pH of the solution after the following volumes of base have been added:
(a) 15.0 mL
(b) 19.9 mL.
115views - Textbook Question
Compare the titration of a strong, monoprotic acid with a strong base to the titration of a weak, monoprotic acid with a strong base. Assume the strong and weak acid solutions initially have the same concentrations. Indicate whether the following statements are true or false. (a) More base is required to reach the equivalence point for the strong acid than the weak acid.
1094views1rank - Textbook Question
Predict whether the equivalence point of each of the following titrations is below, above, or at pH 7: (b) NH3 titrated with HCl.
379views - Multiple ChoiceA 100.0 mL sample of 0.18 M HClO4 is titrated with 0.27 M LiOH. Determine the pH of the solution after the addition of 30.0 mL of LiOH.555views