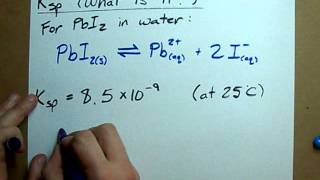18. Aqueous Equilibrium
Solubility Product Constant: Ksp
18. Aqueous Equilibrium
Solubility Product Constant: Ksp
Additional 7 creators.
Learn with other creators
Showing 10 of 10 videos
Practice this topic
- Multiple ChoiceDetermine the molar solubility of CaCO3 in a solution of 0.500 M Na2CO3. Ksp for CaCO3 is 4.96 × 10−9.1138views
- Multiple ChoiceWhat is the molar solubility of PbBr2 in pure water? Ksp for PbBr2 is 4.67 × 10−6.1361views
- Multiple Choice
Solubility of Sn(OH)2 was found to be 1.11 x 10-9 M; calculate Ksp of this compound.
1006views9rank - Multiple Choice
If a saturated solution of Ag2CO3 contains 2.56 × 10−4 M of Ag+ ions, determine its solubility product constant.
820views6rank1comments - Textbook QuestionWhat is the solubility-constant expression for Ca3(PO4)2 (s)? (a) [Ca2+]3 [PO43-]2(b) [Ca2+]3 [PO43-]2 / [Ca3(PO4)2](c) [Ca(PO4)2] / [Ca2+]3 [PO43-]2(d) [Ca2+]2 [PO43-]3981views
- Textbook Question
(b) Write the expression for the solubility-product constant for each of the following ionic compounds: MnCO3, Hg(OH)2, and Cu3(PO4)3.
713views - Textbook Question
(a) True or false: 'solubility' and 'solubility-product constant' are the same number for a given compound.
929views - Textbook Question
(a) I f t he molar solubility of CaF2 at 35°C i s 1.24 × 10–3 mol/L, what is Ksp at this temperature?
1077views1rank