17. Acid and Base Equilibrium
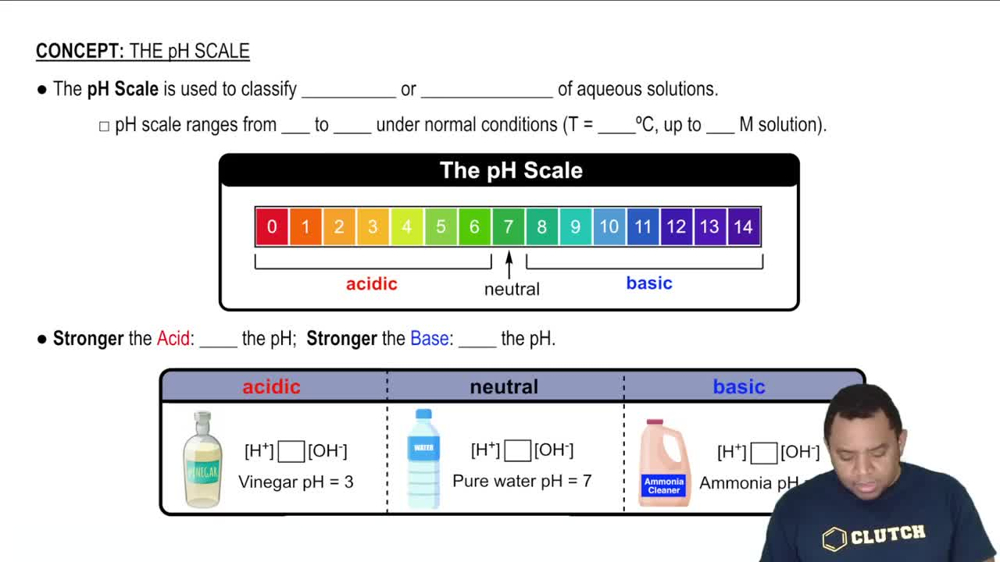
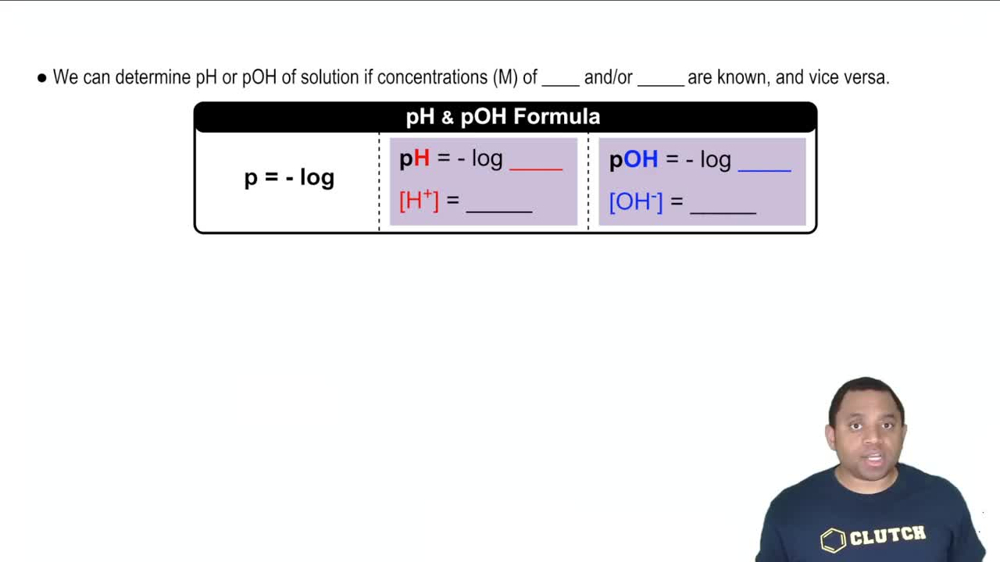
The pH Scale
Learn with other creators
Practice this topic
- Multiple Choice
Which of the following solutions will have the lowest concentration of hydronium ions?
1773views2rank6comments - Multiple Choice
Which of the following statements about aqueous solutions is/are true?
5023views3rank - Multiple Choice
What is the Kw of pure water at 20.0°C, if the pH is 7.083?
4695views7rank5comments - Multiple Choice
Calculate the pH of 50.00 mL of 4.3 x 10-7 M H2SO4.
3025views4rank5comments - Textbook Question
The probe of the pH meter shown here is sitting in a beaker that contains a clear liquid. (c) Why is the temperature given on the pH meter?
358views1rank - Textbook QuestionWhat is the concentration of hydroxide ions 3OH-4 in a glass of wine with pH = 3.64? (LO 16.5, 16.6) (a) 2.3 * 10-4 M (b) 6.4 * 10-3 M (c) 6.8 * 10-9 M (d) 4.4 * 10-11 M477views
- Textbook Question
Calculate [H+] for each of the following solutions, and indicate whether the solution is acidic, basic, or neutral: (a) [OH-] = 0.00045 M (b) [OH-] = 8.8 × 10-9 M (c) a solution in which [OH-] is 100 times greater than [H+].
375views - Textbook Question
Calculate 3OH-4 for each of the following solutions, and indicate whether the solution is acidic, basic, or neutral: (b) 3H+4 = 2.5 * 10-10 M
394views