17. Acid and Base Equilibrium
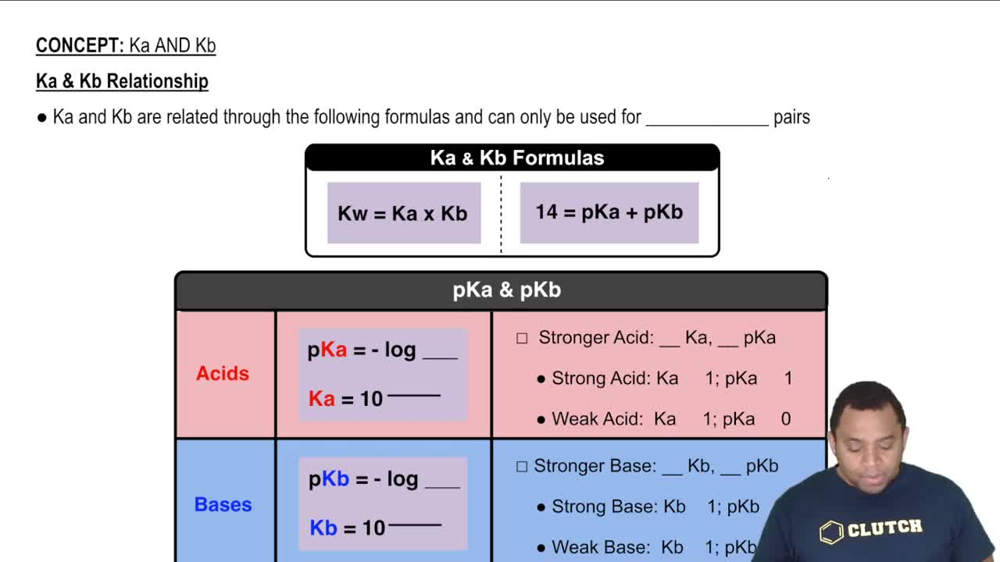
Ka and Kb
17. Acid and Base Equilibrium
Ka and Kb
Additional 3 creators.
Learn with other creators
Showing 6 of 6 videos
Practice this topic
- Multiple Choice
If the Kb of NH3 is 1.76 x 10-5, determine the acid dissociation constant of its conjugate acid.
3826views1rank3comments - Multiple Choice
Which Bronsted-Lowry base has the greatest concentration of hydroxide ions?
2870views2rank - Multiple Choice
Which Bronsted-Lowry acid has the weakest conjugate base?
4242views4rank1comments - Multiple ChoiceThe Ka value for a weak acid is _________ than the Ka value for a strong acid because the weak acid ionizes to a _________ extent than the strong acid.1050views
- Open QuestionWhat is the 𝐾a reaction of hcn?927views
- Open Question
What is Kb for the conjugate base of HCN (Ka = 4.9 × 10-10)?
650views - Open Question
What is Kb for the conjugate base of HCN (Ka = 4.9 × 10−10)?
725views - Open Question
Given that Ka for HIO is 3.2×10−11 at 25 °C, what is the value of 𝐾b for IO− at 25 °C?
676views