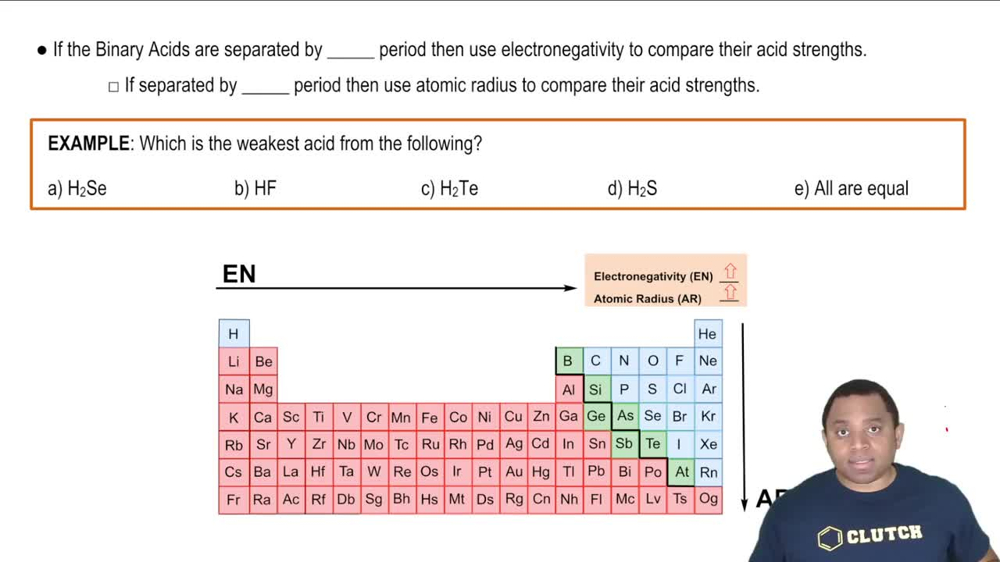
17. Acid and Base Equilibrium
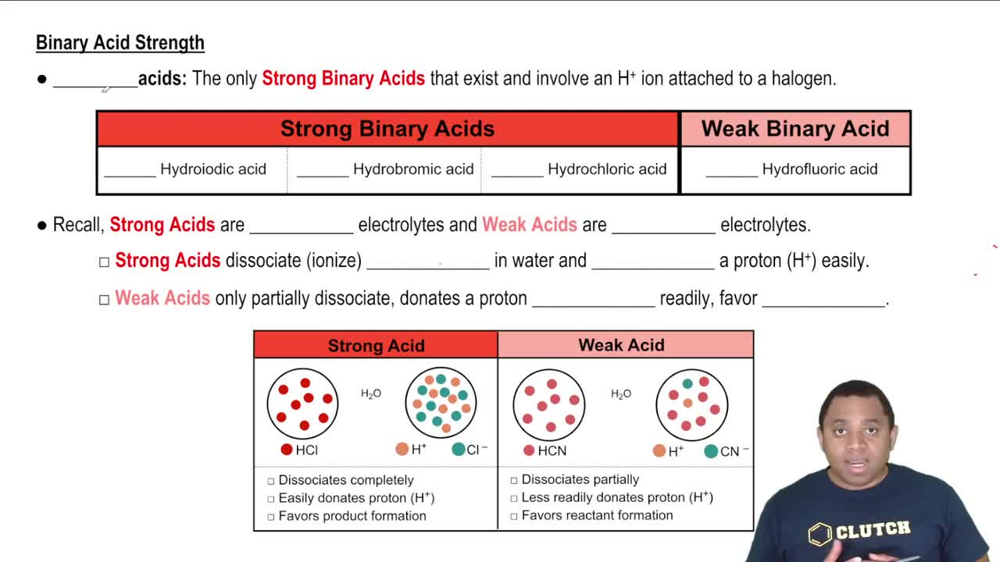
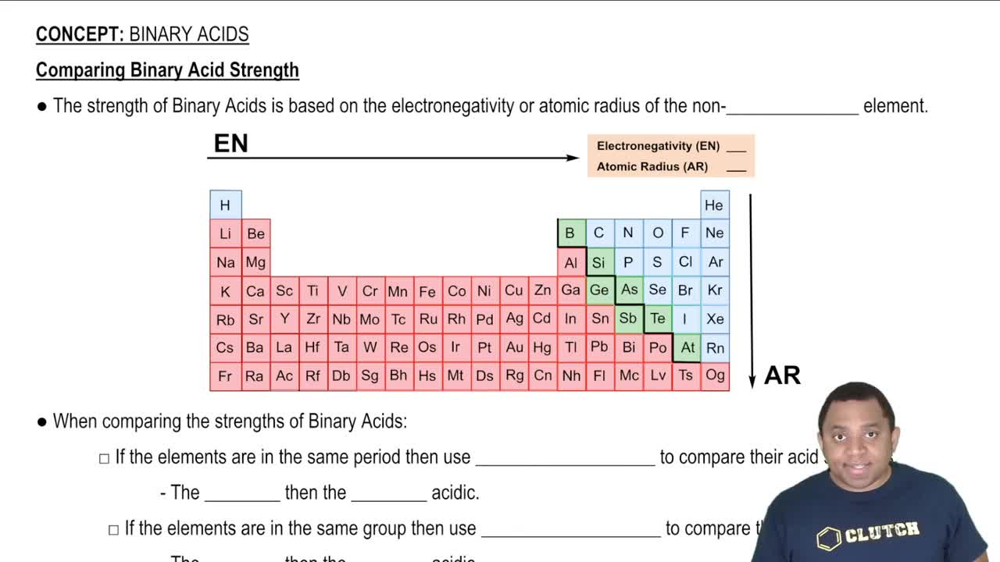
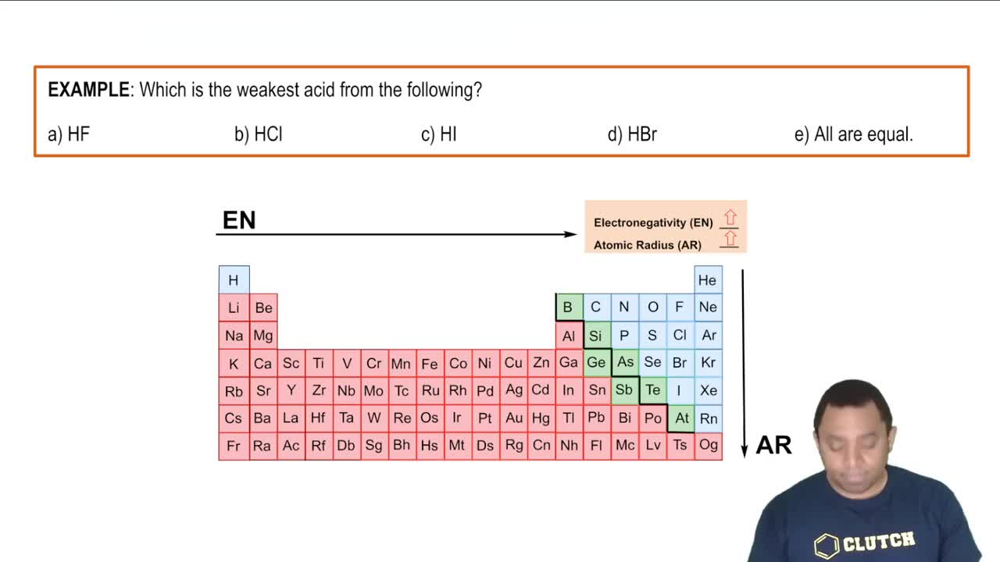
Binary Acids
17. Acid and Base Equilibrium
Binary Acids
Showing 7 of 7 videos
Practice this topic
- Multiple Choice
Which of the following acids would be classified as the strongest?
a) CH4 b) H2Te c) H2S d) PH3 e) BH3
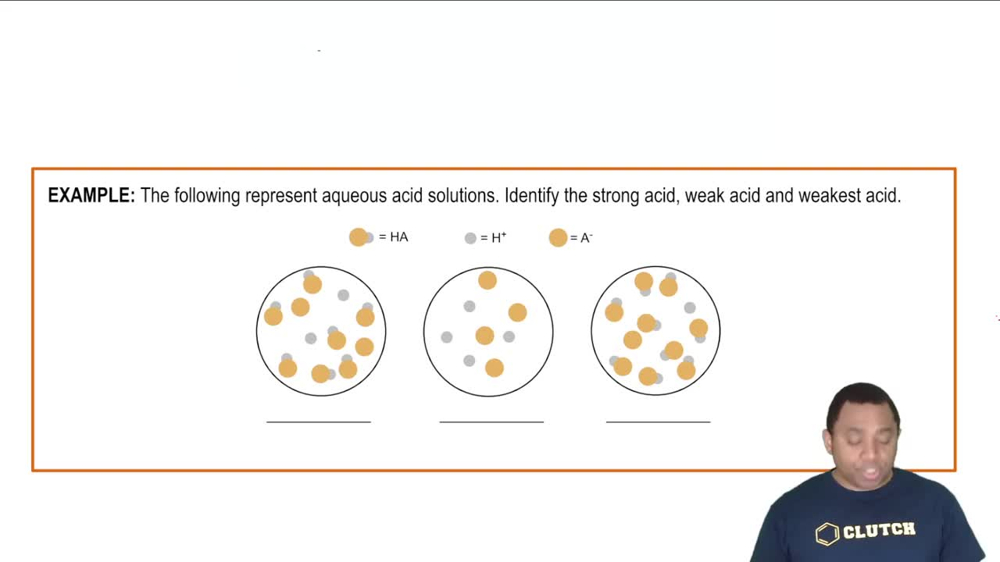
905views7rank4comments - Textbook QuestionThe following pictures represent equal volumes of aqueous solutions of three acids HA (A = X, Y, or Z); water molecules have been omitted for clarity. Which is the strongest acid?(a) HX (b) HY (c) HZ (d) All three acids are strong acids and have equal strength.704views
- Textbook Question
The following diagrams represent aqueous solutions of three acids, HX, HY, and HZ. The water molecules have been omitted for clarity, and the hydrated proton is represented as H+ rather than H3O+.(c) Which solution would have the highest pH?
1941views1rank - Textbook QuestionWhich of the following solutions is the most acidic? (a) 0.2 M Ba(OH)2, (b) 0.2 M H2SO3, (c) 1.0 M glucose 1C6H12O6).1557views2rank
- Textbook Question
State whether each of the following statements is true or false. Justify your answer in each case. (b) HCl is a weak acid.
329views - Multiple ChoiceBinary acids are formed when hydrogen bonds with a single nonmetal. What is the chemical formula for hydroiodic acid?18views
- Multiple ChoiceWhat is the chemical formula for hydrosulfuric acid?26views
- Multiple ChoiceWhich binary acid is primarily responsible for the acidity of gastric juice in the human stomach?25views