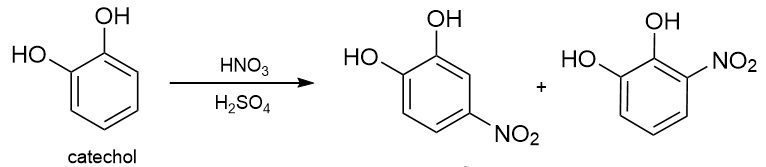
Before modern spectroscopic techniques, Körner’s approach was employed to distinguish between the ortho, meta, and para isomers of disubstituted benzene derivatives. This method involves introducing a third substituent (commonly a nitro group) and determining the number of isomeric products formed. For instance, when catechol undergoes nitration, two distinct isomers are obtained.
An early 20th-century chemist isolated a disubstituted benzene with the molecular formula C6H4Cl2. Upon careful nitration and subsequent purification, three isomeric derivatives with the formula C6H3Cl2NO2 were obtained. Propose a plausible structure for the original compound and the three nitrated isomeric products.