Consider the structures of the four bases [guanine-cytosine and adenine-thymine pairs]:
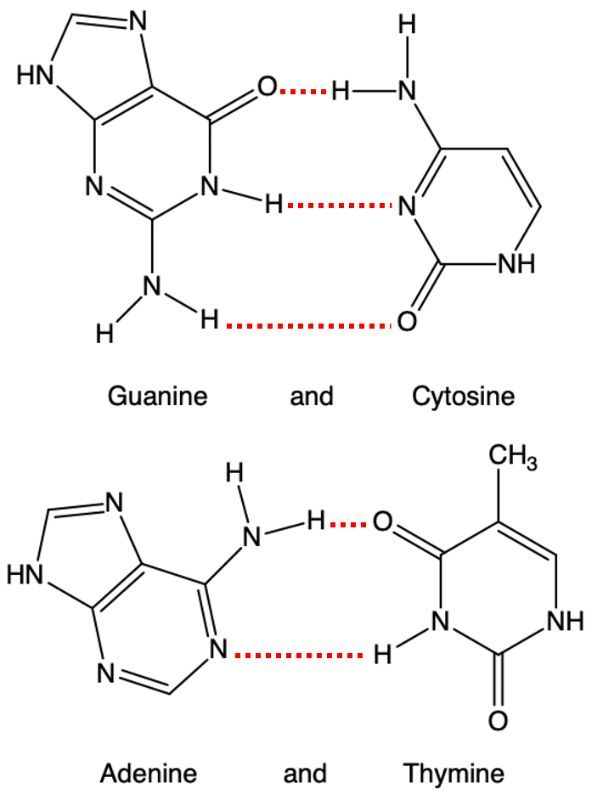
(i) Draw the polar resonance forms of the amide groups [R(C=O)NR'R''].
*Note that a C connected to an O and a N is a region of low electron density.
(ii) Show how the polar resonance forms help to justify why the H-bonds involved in these pairings are strong.
*Note that a H-bond arises between an electron-rich pair of nonbonding electrons and an electron-deficient H atom.