The majority of amino acids found in nature are L-amino acids, whereas D-amino acids are infrequently present in natural proteins. Chemists occasionally need to synthesize D-amino acids as components of drug or insecticide synthesis. One method for generating D-amino acids involves converting the corresponding L-amino acids. The following demonstrates the process of synthesizing D-isoleucine from L-isoleucine.
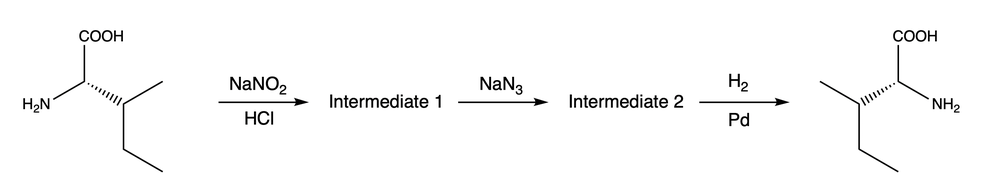
i. What are the structures of the missing intermediates 1 and 2?
ii. How can we verify that the aforementioned synthesis yields the D-configuration that is not found in nature?