6. Chemical Quantities & Aqueous Reactions
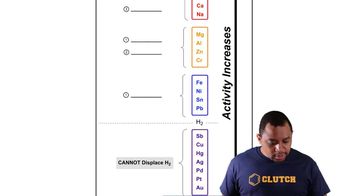
Activity Series
Problem 105d
Textbook Question
Textbook QuestionSuppose you have 3.00 g of powdered zinc metal, 3.00g of powdered silver metal and 500.0 mL of a 0.2 M copper(II) nitrate solution. (d) What is the molarity of Cu2+ ions in the resulting solution?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
12mPlay a video:
347
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos