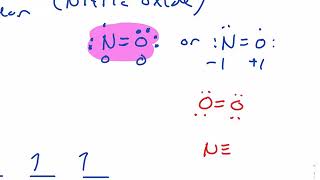12. Molecular Shapes & Valence Bond Theory
MO Theory: Heteronuclear Diatomic Molecules
Problem 111
Textbook Question
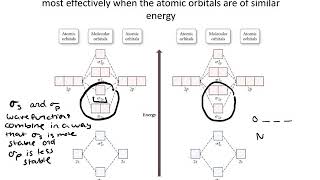
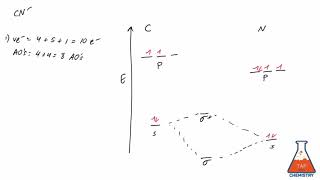
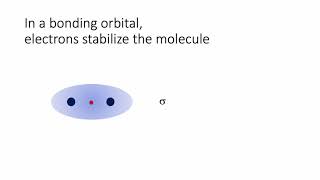
Textbook QuestionCarbon monoxide, CO, is isoelectronic to N2. (d) Would you expect the p2p MOs of CO to have equal atomic orbital contributions from the C and O atoms? If not, which atom would have the greater contribution?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1202
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos