18. Aqueous Equilibrium
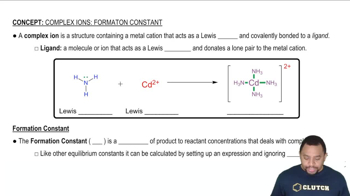
Complex Ions: Formation Constant
Problem 65
Textbook Question
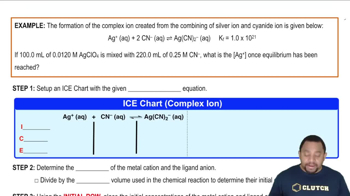
Textbook QuestionFrom the value of Kf listed in Table 17.1, calculate the concentration of Ni2 +1aq2 and Ni1NH326 2+ that are present at equilibrium after dissolving 1.25 g NiCl2 in 100.0 mL of 0.20 M NH31aq2.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
872
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos