10. Periodic Properties of the Elements
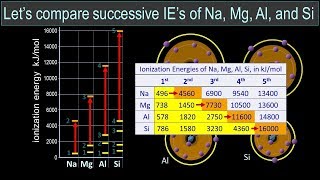
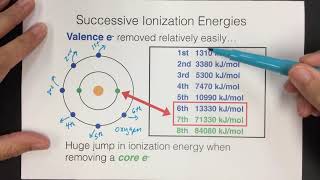
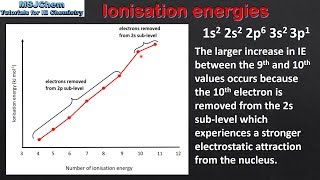
Periodic Trend: Successive Ionization Energies
Problem 77
Textbook Question
Textbook QuestionFor each element, predict where the 'jump' occurs for successive ionization energies. (For example, does the jump occur between the first and second ionization energies, the second and third, or the third and fourth?) a. Be b. N c. O d. Li
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
3100
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos