18. Aqueous Equilibrium
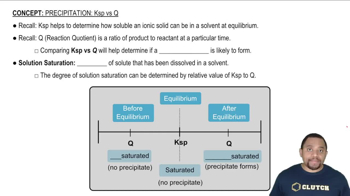
Precipitation: Ksp vs Q
Multiple Choice
Multiple ChoiceTwo mixtures are added into one flask at 25 °C, one mixture contains 0.55 mL of 0.75 M BaF2 and another 0.25 mL of 1.3 M Mg(OH)2. Ksp of Magnesium Fluoride, MgF2, is 7.4 x 10−9. Identify the correct option.
a) MgF2 solid will form
b) MgF2 solid forms, along with Mg+2 and F− ions
c) solution is unsaturated, precipitate does not form
d) solution is saturated, precipitate forms
A
MgF2 solid will form
B
MgF2 solid forms, along with Mg+2 and F− ions
C
solution is unsaturated, precipitate does not form
D
solution is saturated, precipitate forms
170
views
1
rank
1
comments
Related Videos
Related Practice
Showing 1 of 2 videos