11. Bonding & Molecular Structure
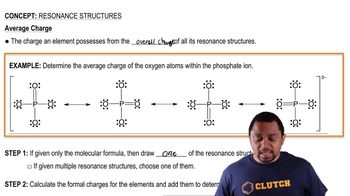
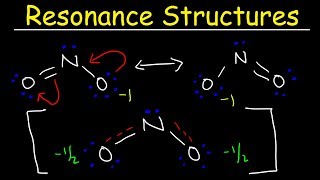
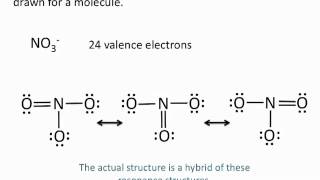
Resonance Structures
Problem 68b
Textbook Question
Textbook QuestionSome chemists believe that satisfaction of the octet rule should be the top criterion for choosing the dominant Lewis structure of a molecule or ion. Other chemists believe that achieving the best formal charges should be the top criterion. Consider the dihydrogen phosphate ion, H2PO4-, in which the H atoms are bonded to O atoms. (b) What is the predicted dominant Lewis structure if achieving the best formal charges is the top criterion?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
550
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos